
Carbon Tetrachloride can be expressed as CCl4, and it is made out of one carbon molecule and four chloride molecules. Carbon tetrachloride is nonpolar. Why is carbon tetrachloride nonpolar? It is nonpolar because the dipole moments of the molecule are evenly spaced around the central carbon atom. This means that their individual effects are canceled out, and the molecule is neutral.
The four chlorine atoms are positioned symmetrically at the four corners of a tetrahedron, and a single bond joins each of them to the carbon atom in the center of the molecule. As a result, CCl4 has no net positive or negative dipole moment. If an atom other than chlorine had been positioned at any other place in a molecule, the molecule would have a dipole moment and CCl4 would be polar as a result.
“We define organic chemistry as the chemistry of carbon compounds.” — August Kekule
That’s the quick answer regarding the polarity of carbon tetrachloride. However, it would be beneficial to talk about the polarity of molecules in general and examine carbon tetrachloride and its properties to understand why it is nonpolar in nature.
What Does It Mean For A Molecule To Have Polarity?
When you hear the term polar, you likely think of the north and south poles on the earth. These north and south poles are located at opposite ends of the earth, similar to how a battery can have a positive and negative pole. Molecules and the bonds between atoms can also have polarity. A molecule is defined as polar when the atoms that constitute the molecule are arranged in a way that gives one end of the molecule a positive charge and the other end of the molecule a negative charge.
A polar molecule is created when an atom that has a high electronegativity level combines or bonds with an atom that has a weaker electronegativity level. The resulting molecule has one region with high electronegativity and one region with lower electronegativity or electrical poles. One of the most famous examples of a polar molecule is water, and it is thanks to water’s polar nature that it serves as the basis for life on earth.
In contrast, nonpolar molecules lack electrical poles, and the electrons within them are distributed in a more equal fashion. Nonpolar molecules lack a notable charge at either end of the molecule. Most hydrocarbon liquids are nonpolar in nature.
In sum, a molecule that has dipoles that do not cancel each other out is polar, whereas nonpolar molecules are those where the charges of atoms cancel one another out and there is no charge at one end of the molecule or the other.
Examples Of Polar Molecules
As previously mentioned, water is a polar molecule. The bonds that exist between the oxygen atoms and hydrogen atoms are distributed such that both sides of the atom, both of the oxygen-hydrogen and bonds, are evenly spaced. The result is that the oxygen side, or top of the molecule, has a slightly negative charge while the hydrogen atoms, or the bottom of the molecule, has a slightly positive charge.
Ethanol is a polar molecule because the oxygen atoms that are within the molecule have more electronegativity than the other atoms. The oxygen atoms attract more electrons due to their higher electronegativity potential, and the -OH bond groups in the molecule have negative charges overall, even if the negative charge is a very slight one.
“Life exists in the universe only because the carbon atom possesses certain exceptional properties.” — James Jeans
Other examples of polar molecules include sulfur dioxide (SO2), hydrogen sulfide (H2S) and ammonia (NH3).
Be warned that a molecule can be comprised of polar bonds and yet still be nonpolar. An example is carbon dioxide, which has four bonds yet the dipole moments of the molecule end up neutralizing one another so the molecule is nonpolar.
Examples Of Nonpolar Molecules

Skeleton formula of methane. Photo: By Jynto – Own work, based on File:Methane-CRC-MW-dimensions-2D.png, Public Domain, https://commons.wikimedia.org/w/index.php?curid=12422898
Examples of nonpolar molecules include nitrogen, methane, ozone, and oxygen. These are homonuclear molecules or molecules composed of atoms of only one element. Nonpolar molecules that aren’t homonuclear include carbon dioxide, mentioned above and methane. Toluene and gasoline are examples of nonpolar substances as well. As a general rule, carbon compounds are nonpolar, though there are some exceptions such as carbon monoxide. Carbon monoxide is linear in nature, which would usually make a molecule nonpolar, yet the electronegativity difference between the oxygen and carbon molecule is substantial enough that the molecule is polar. Alkynes don’t dissolve in water, and they are considered nonpolar molecules. The inert or noble gases are also classified as nonpolar molecules because the gases are only single atoms of their respective elements such as neon, helium, argon, and krypton.
Predicting The Polarity Of A Molecule
Examining the electronegativity values of the atoms in a molecule will help you determine whether or not the molecule is likely to be polar or nonpolar. Significant differences between electronegativity values imply that the electrons are equally shared between the atoms in the molecule. Since the electrons will be closer to one atom than to another, that region of the molecule will be polar, though to determine the polarity of the entire molecule all bonds must be taken into account.
The deciding factor in a molecule’s polarity or non-polarity is its geometry. If one end of the molecule has a negative charge while the other end of the molecule has a positive charge, the molecule will be polar. Yet if charges are evenly distributed and orbit a central atom, the molecule is probably nonpolar. You can use this to predict whether a molecule will be polar or nonpolar. Not every molecule has a dipole moment, and this can make predicting the polarity of a molecule a little more difficult. As an example, molecules that can be flipped and mirrored across a geometric plane, don’t have dipole moments because dipole moments can’t comprise more than a single point.
Facts About Carbon Tetrachloride
Carbon tetrachloride was first discovered by the French chemist Henri Victor Regnault in the mid-1800s. The chemical was discovered through a combination of chlorine and chloroform. Despite this, carbon tetrachloride is mainly derived from methane nowadays. The production of carbon tetrachloride frequently operates by using the byproducts of chlorination reactions to derive the compound. This includes the byproducts of the creation of chloroform and dichloromethane. Carbon tetrachloride is fairly volatile in nature, giving off a smell similar to dry cleaning chemicals. Carbon tetrachloride is also solvent and therefore is useful in the dissolving of nonpolar substances like oils and fats.
Carbon tetrachloride has many different applications and has been used as a cleaning agent, as a refrigerant, and in fire extinguishers. Carbon tetrachloride is often used in the creation of lava lamps, making the wax heavier.
“… Is the carbon molecule lined with thought?” — Saul Bellow
Because of the environmental impact of carbon tetrachloride, its production has steeply declined since the 1980s. The Montréal protocol limited the production of chlorofluorocarbons, and since carbon tetrachloride is a key ingredient in the creation of CFCs, carbon tetrachloride has seen a sharp decrease in production as a result. The adverse health effects of carbon tetrachloride have also caused a decrease in its production, and though it was once widely used as a solvent, It is rarely used for such purposes nowadays.
Carbon tetrachloride is extremely toxic to the liver, and other liver-damaging substances are often compared against the toxicity of carbon tetrachloride. High concentrations of carbon tetrachloride can not only damage the liver, but they can also damage the kidneys. Prolonged exposure to carbon tetrachloride can cause a coma or death. Exposure to the chemical could also increase one’s chance of developing cancer.
Why Is Carbon Tetrachloride Polar?
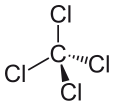
Photo: NEUROtiker via Wikimedia Commons, Public Domain
After seeing what makes a molecule polar, and looking at some examples of polar molecules, we can now better understand why carbon tetrachloride is a nonpolar molecule. The first thing that should be done when trying to determine if a molecule is polar and nonpolar is to calculate the electronegativity values of the atoms within the molecule. The polarity of the bond between two atoms must be ascertained, and all bonds must be considered when trying to determine the polarity of the molecule. Yet the difference of electronegativity values between chloride and carbon, in the case of carbon tetrachloride, isn’t the only factor that must be considered. The shape of the molecule must be considered as well. While there are dipoles present within the molecule, the linear shape of carbon tetrachloride means that the effects of the different dipole moments in the molecule cancel one another out, and the molecule is neutral or nonpolar as a result.









