
With the ever-increasing demand for cleaner energy, natural gas is playing a growing role in the global energy mix replacing coal and oil to help transition into a future carbon-constrained world. Methane hydrates (MHs) have been considered as the potential future resource of energy due to its abundant resource volume of CH4 (20,000 trillion cubic meter) in nature.
MHs are stable at suitable low temperatures and high-pressure conditions. They are typically found at permafrost locations and offshore locations near the continental shelf. This resource volume is probably twice of the total oil and gas reserves on earth. Exciting R&D programs are ongoing in Japan, China, Korea, India, and the U.S.A., trying to exploit the potential of MHs for energy within the next 20 years.
(Image republished with permission)
The numerical simulation of the production performance of MH reservoirs under different production techniques is an important tool, as it provides critical information on the production rate and duration of reservoir fluids (water and CH4 gas). It is practically the only method that is able to predict long-term production from MH reservoirs since most field tests are short in duration because of the hostile production environment and high capital investment.
To tackle this front, the state-of-the-art reservoir simulator of methane hydrate (TOUGH+Hydrate v1.5) was developed by the Lawrence Berkley National Laboratory to model the dynamic behavior of MH formation and dissociation together with the associated fluids production behavior in geological media accounting for all known physics (thermodynamics, heat transfer, fluids flow, phase change criteria). Such reservoir simulator tools though powerful in its efficiency, require validation against laboratory experimental data or field production data to install confidence in its predictions.
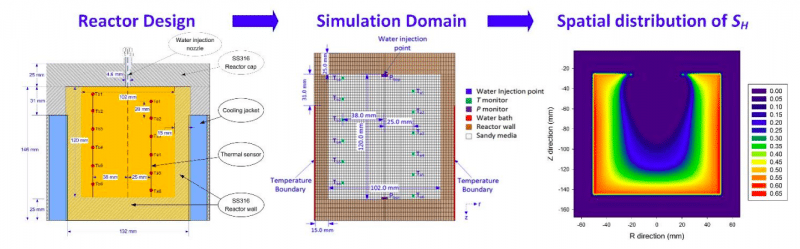
In this direction, a recent study faithfully duplicated the multi-stage MH formation process in an unconsolidated sandy medium in a 1.0 L reactor numerically by employing the T+H v1.5 simulator based on a history-matching technique. Simulation prediction of the system performance (pressure and temperature) achieved excellent agreement with the experimental observation. Their study reveals that the phase change behavior of MH formation and dissociation in the sandy porous medium is better represented by a kinetic reaction model rather than an equilibrium reaction model.
Moreover, from the simulation prediction, it is elucidated that the spatial distribution of hydrate (SH) in sandy medium inside reactor can be significantly heterogeneous varying from SH = 5% in the reactor center to SH = 65% towards the reactor boundary. This finding is a breakthrough, which improves our understanding of the process of MH formation both in terms of temporal and spatial distribution.
These findings are described in the article entitled Numerical analysis of experimental studies of methane hydrate formation in a sandy porous medium, recently published in the journal Applied Energy. This work was carried out by Zhenyuan Yin, George Moridis, Hoon Kiang Tan and Praveen Linga. This is a collaboration work involving the Department of Chemical & Biomolecular Engineering in the National University of Singapore, Lawrence Berkeley National Laboratory, and Lloyd’s Register Singapore Pte. Ltd.








