
Along with the dramatically shrinking amount of petroleum and growing energy consumptions, an increasing number of individuals focus on coal conversion into chemicals and liquid fuels. Lignite is an abundant fossil resource found in nature, but in spite of its abundance, easy access, and low mining cost, it has not been used as widely as high-rank coal because of its high moisture content, lower calorific value, high spontaneous combustion susceptibility, and high ash yield.
The conditions of traditional coal chemical technology processes are relatively harsh, which limits the coal’s high value-added utilization. However, the catalyst can convert the coal under mild conditions to achieve clean utilization. Therefore, more and more researchers have shifted their focus to obtaining highly active, selective catalysts.
Lignite is rich in oxygen-containing bridged bonds, especially the C−O bridged bonds (COBBs), such as BRCH2O–CH2BR, which are the predominant COBBs in lignite. Therefore, effectively cleaving these bridged bonds should be crucially important for coal’s utilization. Solid superbase is considered to be effective for catalyzing the cleavage of COBBs because of their strong basicity, resulting in heterolytically splitting H2 to release H–.
One of the main challenges in practical applications of the heterogeneous solid catalysts in hydrocracking of coals is that the difficulties in their separation from the reaction mixture and subsequent recycling. Therefore, magnetic ferroferric oxide nanoparticles have attracted considerable interests as catalyst carriers because of their superparamagnetic property, which makes the catalysts easily separated by an external magnetic field.
A highly active magnetic solid superbase (MSSB) was prepared by impregnating Mg2Si onto γ-Al2O3 coated ferroferric oxide nanoparticles via one-pot synthesis method and can be flexibly separated from the reaction mixture by an external magnetic field. MSSB successfully applied in the catalytic hydroconversion (CHC) of Oxybis(methylene) dibenzene (OBMDB) and the extraction residue (ER) from Naomaohu lignite.
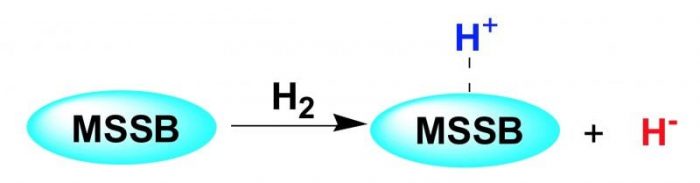
Scheme 1. Proposed pathways for the formation of H– over MSSB. Republished with permission from Elsevier from https://doi.org/10.1016/j.fuel.2018.03.019.
OBMDB was used as a lignite-related model compound to evaluate the catalytic activity of MSSB contains oxygen-containing bridged linkage (i.e., –CH2OCH2–) between 2 benzene rings. Generally, the conversion of OBMDB along with their main products monotonously increased with further raising the reaction temperature, prolonging the reaction time, increasing the reaction initial hydrogen pressure (IHP), and increasing the MSSB dosage, respectively.
This study explored the possible mechanism for the CHC of OBMDB. Unlike the acidic catalyst, solid base catalysts can facilitate heterolytically splitting H2 to release H– and subsequent H– transfer because of its strong alkalinity. Due to the strong electronegativity of oxygen atom in OBMDB, the two carbon atoms connected to oxygen atom are positively charged and thereby tend to accept H–.
The mobile H– transfers to the oxygen atom in OBMDB and subsequent the two same COBBs are cleaved to yield toluene and BRCH2O–. BRCH2O– abstracts H+ from MSSB surface to produce benzalcohol or transforms into benzaldehyde. Due to its poor stability, benzalcohol dehydroxylation proceeds via H– transfer to the benzylic carbon in benzalcohol to produce toluene and HO–, HO– can abstract H+ from MSSB surface to produce H2O. Moreover, OBMDB cannot react without IHP, and benzalcohol was only detected under 1 MPa IHP, indicating that IHP plays an important role in the CHC process.
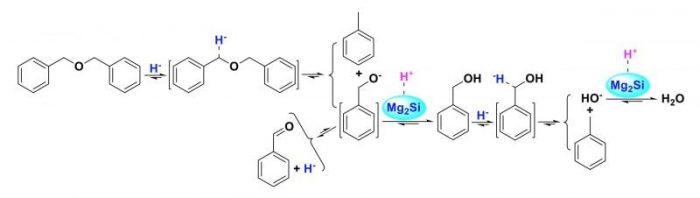
Scheme 2. Proposed pathways for OBMDB hydrocracking over MSSB. Republished with permission from Elsevier from https://doi.org/10.1016/j.fuel.2018.03.019.
Under optimal conditions, MSSB is applied in the CHC of the ER. Non-catalytic hydroconversion (NCHC) were reacted under the same conditions as CHC except for not added catalyst. The final reside from the CHC (FR), ER, SPNCHC, and SPCHC were analyzed with a Nicolet Magna IR-560 Fourier transform infrared (FTIR) spectrometer, and the filtrate was analyzed with an Agilent 7890/5973 gas chromatograph/mass spectrometer (GC/MS). Results revealed that the SPNCHC contains more alkanes and arenes, while the SPCHC have more alkenes and oxygen-containing compounds.
It is interesting that only 8 kinds of group components were detected in the SPNCHC. However, 17 kinds of group components exist in the SPCHC. It is indicated that the CHC is beneficial to the fracture of C–O bond and the production of phenolic and ester compounds. The total relative content (TRC) of phenols in the SPCHC is significantly higher than that in the SPNCHC, in which most of the phenols contain a benzene ring. In contrast, the TRCs of alkanes and arenes in the SPCHC are appreciably lower than those in the SPNCHC.
These results indicate that most of the COBBs in the ER were hydrocracked over MSSB. In other words, MSSB was proved to be highly active for the CHC of macromolecular moieties in the ER to form phenols and alcohols. The higher TRC of phenols in the SPCHC than in the SPNCHC is consistent with the result of FTIR analysis.
These findings are described in the article entitled Catalytic hydroconversion of the extraction residue from Naomaohu lignite over an active and separable magnetic solid superbase, recently published in the journal Fuel. This work was conducted by Min Zhang, Xian-Yong Wei, Zheng Yang, Dao-Guang Teng, Yan Xue, De-Wu Meng, and Zhi-Min Zong from the China University of Mining & Technology.









