Osteoporosis is a skeletal disorder that predisposes patients to an increased risk of bone fractures. Osteoporosis is very common among elderly women and — to a lesser extent — men. Worldwide, 1 in 3 women and 1 in 5 men over the age of 50 will experience an osteoporotic fracture, which entails immobilization, further weakening, and elevated risk of additional fractures (Melton LJ, 3rd et al. J Bone Miner Res 1998; Melton LJ, 3rd et al. J Bone Miner Res Kanis JA et al. Osteoporos Int).
Fractures due to osteoporosis account for approximately 1% of the disability and premature mortality attributable to non-communicable diseases, taking a huge personal and economic toll (Hernlund et al. Arch Osteoporos 2013). Therefore, it is of utmost interest to identify patients at risk early enough to begin with pharmacologic and lifestyle interventions to avoid that first fracture. Available medication can lower fracture risk by up to 70%, however, currently applied methods for fracture risk assessment based on bone mineral density measured by Dual Energy X-ray Absorptiometry (DXA), or on scoring of clinical risk factors by questionnaires like FRAX®, have limited utility for identifying patients in urgent need of therapeutic interventions.
MicroRNAs are small RNA molecules that control changes in cell function by selectively regulating the synthesis of proteins. Circulating microRNAs, which are secreted into the blood by many cell types, are associated with physiologic and pathologic changes in the body and thus may allow conclusions on the development and progression of diseases before disease symptoms are evident.
We have investigated the role of microRNAs in the context of bone function and dysfunction and have found that concentrations of certain microRNAs differ between fractured and non-fractured patients of osteoporosis (Kocijan et al., J Clin Endocrinol Metab. 2016; Heimeier et al., J Bone Miner Res. 2016), are responsive to anti-resorptive treatment, and are associated with parameters of bone quality as detected by imaging techniques (Feichtinger et al., Sci Rep 2018). Based on our research findings, we have developed the osteomiR™ test, a fracture risk assessment tool based on microRNAs measured in blood samples. In order to generate evidence of the test’s usefulness in terms of benefit to the patient and resource consumption, and to compare its usefulness with that of the standard of care, we have conducted a health-economic evaluation.
Health economics is an interdisciplinary branch of science that studies the functioning of healthcare systems and health-affecting behaviors. For example, health economists evaluate the cost and outcomes of a pharmaceutical product in order to enable optimal healthcare resource allocation based on scientific evidence. One frequently-applied method in health economic evaluation is cost-utility analysis, which puts the cost of a health-related intervention in relation to its benefit in terms of quality of life.
In the present article, we describe how we used mathematical modeling to conduct a cost-utility analysis. We simulated long-term consequences of fracture risk assessment and resulting treatment decisions using the osteomiR™ test in comparison with DXA, FRAX® or no preventive examination. According to the expected test results, the model assigns each individual to a risk group (low, middle, high) with a given accuracy. Individuals assigned to the high-risk group receive medicinal treatment, which lowers fracture risk.
Depending on the test accuracy, the proportion of individuals receiving treatment and, consequently, the number of occurring fractures will vary, as will costs related with medical screening, medicinal treatment, and fracture care. As many factors influencing cost and outcomes (like e.g. disease prevalence, fracture probabilities, pricing of medical screening, fracture care, and pharmacological treatments) depend on local circumstances, we have based our analysis on the actual conditions in Austria. All numerical data entering the model are derived from meticulous literature research (costs, probabilities, predictive accuracy of DXA and FRAX®) or from our own research (predictive accuracy of osteomiR™). Finally, sensitivity analyses were performed to assess how variations of individual input parameter values affect the results.
According to our model, fracture risk assessment and monitoring using the osteomiR™ test reduces fracture incidence compared with no monitoring, DXA and FRAX®, due to its superior capacity to select patients requiring therapeutic intervention. Considering the total cohort over a lifetime, nationwide application of the osteomiR™ test can avoid 57,919 fractures compared with DXA, 31,285 fractures compared with FRAX® and 133,394 fractures compared with no screening/monitoring.
When considering the cost associated with each screening method, we found only small variations in the total cost per patient: Application of the osteomiR™ test results in slightly enhanced costs for medication, as more patients are selected for medicinal treatment, but this is balanced by reduced costs for fracture care. When compared to DXA, each additional year that is spent in full health thanks to osteomiR™, is associated with a saving of 19,605 €! Such medical interventions that help to gain quality of life while at the same time maintaining or even reducing resource consumption, are clearly highly desirable. Our results, therefore, recommend implementation of microRNA-based osteoporosis management into clinical practice.
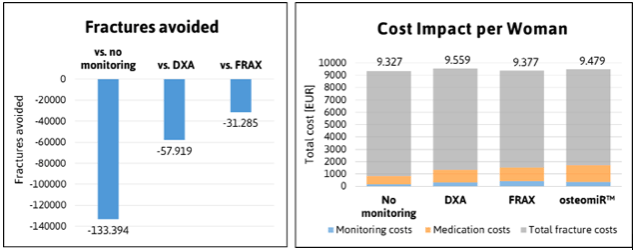
Left: Impact of osteomiR™ utilization on fracture incidence. Right: Total cost per woman over a lifetime are shown in EUR. Uptake of the osteomiR™ test increases medication and monitoring cost while decreasing fracture cost. Credit: Hanna Dellago
In sum, our simulations provide evidence for the utility of the osteomiR™ test for fracture risk assessment, and, furthermore, highlight the general potential of circulating microRNAs to be exploited as minimal-invasive diagnostic tools.
These findings are described in the article entitled Cost-utility analysis of fracture risk assessment using microRNAs compared with standard tools and no monitoring in the Austrian female population, recently published in the journal Bone.
This work was conducted by Evelyn Walter from the Institute for Pharmaeconomic Research, Hanna Dellago and Matthias Hackl from TAmiRNA GmbH, Johannes Grillari from the University of Natural Resources and Life Sciences Vienna, and Hans Peter Dimai from the Medical University of Graz.









