
Capsaicin (trans-8-methyl-N-vanillyl-6-nonenamide) is the compound in chili peppers (Capsicum annuum) responsible for their “hot” taste. Capsaicin binds as a ligand to the plasma membrane protein, termed transient receptor potential vanilloid-1 (TRPV1), which functions as a nonselective cation channel allowing the influx of Na+ and Ca2+ ions into the cells after stimulation. The TRPV1 protein has, therefore, been termed the capsaicin receptor. TRPV1 can also be activated by endogenous lipids, noxious temperatures, and acidic solutions. Thus, capsaicin, elevated temperature, and protons induce the same response in TRPV1 expressing cells.
TRPV1 channels are found in the peripheral nervous system, in particular in trigeminal and dorsal root sensory ganglia, in the kidney and in the gastrointestinal tract. TRPV1 channels mediate pain sensation and tissue injury-induced inflammatory thermal hyperalgesia. The TRPV1 channel plays an essential role in pain hypersensitivity which is associated with chronic pain conditions.
To understand the functions of TRP channels, one must identify, on the cellular level, their associated intracellular signaling pathways and molecular targets. The stimulation of these TRP channels frequently induces an influx of Ca2+ ions into the cells and the subsequent activation of protein kinases. These intracellular signal transduction pathways ultimately lead to changes in the gene expression pattern of the cells.
We have discovered that stimulation of TRPV1 channels with capsaicin or other ligands activated transcription mediated by the transcription factor activator protein-1 (AP-1). AP-1 is a dimeric transcription factor, composed of proteins of the c-Jun and c-Fos families. Accordingly, stimulation of TRPV1 channels with capsaicin increased c-Jun and c-Fos biosynthesis. As AP-1 is involved in the regulation of proliferation, transformation, differentiation, and programmed cell death in a tissue-specific manner, we link TRPV1 stimulation to the biological functions of AP-1. These data suggest that the intracellular functions of TRPV1 stimulation may rely on the activation of the stimulus-responsive AP-1 transcription factors.
Our data nicely complement earlier observations that TRPV1 stimulation is accompanied by the activation of the transcription factor cAMP response element binding protein (CREB). Following activation, AP-1 and CREB induce the transcription of delayed response genes. We propose that many biological functions of TRP channels can be explained by the activation of stimulus-responsive transcription factor and their delayed response genes. The proteins encoded by those delayed response genes may be responsible for the biochemical and physiological changes following TRP channel activation.
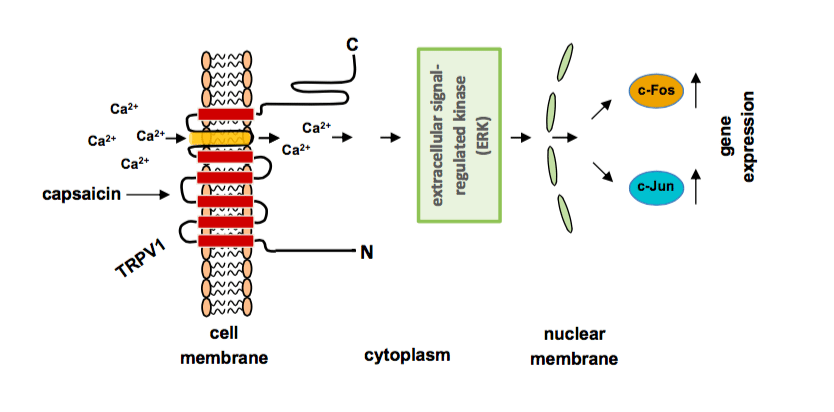
Capsaicin regulates gene transcription: Capsaicin stimultes TRPV1 channels, leading to an influx of calcium ions into the cells and the activation of the extracellular signal-regulated protein kinase ERK. The kinase translocates into the nucleus and stimulates the biosynthesis of c-Jun and c-Fos that both constitute the AP-1 transcription factor. (Image credit: Gerald Thiel)
The fact that TRPV1 stimulation activates AP-1 and CREB suggests the search for those delayed response genes that are activated by these transcription factors following TRPV1 stimulation. A candidate gene is the gene encoding calcitonin gene-related peptide (CGRP) which is regulated by CREB and probably by AP-1 in sensory neurons. CGRP is a neuropeptide that is known to induce neurogenic inflammation. Thus, stimulation of TRPV1 channels triggers an intracellular signaling cascade that leads to the activation of transcription factors which promote the biosynthesis and secretion of a pro-inflammatory peptide.
These findings are described in the article entitled Stimulation of TRPV1 channels activates the AP-1 transcription factor, recently published in the journal Biochemical Pharmacology. This work was conducted by Tobias M. Backes, Oliver G. Rössler, Xin Hui, Peter Lipp, and Gerald Thiel from the Saarland University Medical Faculty, and Carsten Grötzinger from the CharitéCenter.
References:
- Backes, T.M., Rössler, O.G., Hui, X., Grötzinger, C., Lipp, P., Thiel, G. (2018) Stimulation of TRPV1 channels activates the AP-1 transcription factor. Biochem. Pharmacol. 150, 160-169.
- Thiel, G., Lesch, A., Rubil, S., Backes, T.M., Rössler, O.G. (2018) Regulation of Gene Transcription Following Stimulation of Transient Receptor Potential (TRP) Channels. Int. Rev. Cell. Mol. Biol. 335. 167-189.
- Nakanishi, M., Hata, K., Nagayama, T., Sakurai, T., Nishisho, T., Wakabayashi, H., Hiraga, T., Ebisu, S., Yoneda, T. (2010). Acid activation of Trpv1 leads to an up-regulation of calcitonin gene-related peptide expression in dorsal root ganglion neurons via the CamK-CREB cascade: A potential mechanism of inflammatory pain. Mol. Biol. Cell 21, 2568-2577.









