
It is official, the kilogram has been redefined. This change, voted on by the General Conference of Weights and Measures (CGPM) in November 2018, finally came into full force on May 20, 2019, coincident with the 144th anniversary of the first National Metre Convention. Such ends the scientific community’s century-long pursuit for a universal and unchanging system of weights and measurements that will be the standard for scientific measurement for future generations to come.
In a twinge of irony, despite the historical significance of this event, mainstream scientific practice will be virtually unaffected by its coming to fruition. The transition to the new definition of the kilogram will go on unnoticed for most everyday applications. Your bathroom scales won’t change, experimental techniques won’t change, and neither will any fundamental physical equations.
However, this new metric system represents a crowning achievement in the sciences—the goal to have a universal system of units that is not based on human convention or agreement, but that is built right into the fabric of reality itself. The redefinition of the kilogram was the last piece of this puzzle.
What Was the Kilogram Defined As?
Before now, the definition of the kilogram was simple. Since 1889, one kilogram has been defined as exactly the mass of a platinum-iridium alloy cylinder that is kept at the International Bureau of Weights and Measures headquarters in Sevres, France. This object is called the International Prototype Kilogram (also known as “Big K” or “Le Grand K”) For over 100 years, Big K has served as the universal standard for the kilogram, the SI unit for mass. Several copies of Big K exist in research institutions around the world where they are used to calibrate scales and standardize measurements.

A photo of K4 (left) and K20 (right), two copies of the International Prototype Kilogram that were used to standardize measurements in the US. Credit: Public Domain
The main problem with using Big K as a universal standard for mass is that Big K, being a manmade object, is imperfect and subject to change over time. Indeed, it is estimated that Big K has lost about 50 micrograms since the time it was created. Since 1 kilogram is defined as exactly the value of the mass of Big K, if Big K’s mass changes then the value of the kilogram must also change. Obviously, having a standard for mass that changes its mass is not a good idea for science. The ideal set of units should be static and unchanging.
Hence the reason for redefinition. The new definition of the kilogram ties the value of the kilogram to a fundamental constant of nature that will never change. Even billions of years from now when Big K has disintegrated into dust, this new constant the defines the value of the kilogram will be exactly the same—because the value of the unit is tied to a fundamental aspect of reality.
In fact, the kilogram is the last of the base SI units to be defined in terms of some physical constant. Each of the other SI base units (meter, second, ampere, Kelvin, mole, candela) currently have their values defined in terms of some unchanging constant(s) of nature.
The Meter: A Lesson From History
Take the meter for example. From 1799 to 1960, 1 meter was defined as exactly the length of a series of platinum prototype meter bars, exactly how the value of the kilogram was until recently defined by a physical artifact. This physical meter bar, like Big K, was imperfect and subject to change; specifically, temperature changes would cause it to expand and contract.

A close up of National Prototype Meter Bar No. 27, created in 1889. Credit: Public Domain
In 1960, the meter was redefined in terms of the wavelength of the orange emission line spectrum of an atom of krypton-86. This definition was found once again inadequate as it was discovered the orange line emission spectrum was asymmetrical and could be measured as different lengths depending on the chosen point of reference on the line.
In 1983, the definition of the meter was once again changed, this time in reference to the speed of light in the vacuum. The meter was redefined as exactly 1/299,792,458th of the length that a beam of light travels in exactly one second.
The advantage of this new definition is that it is based on the speed of light (denoted with c) which is, as far as we are aware, a universal physical constant. So in other words, the new definition of the meter was built into the definition of the speed of light. The speed of light in a vacuum is the same everywhere in the universe and does not change over time. Thus, our standard for what a meter is will never change. A meter will be the same as it is today 10 billion years from now.
This is the exact same reasoning that motivated the redefinition of the kilogram. Big K is a physical artifact and so will change over time. What scientists needed was a way to define the value of a kilogram with reference to some physical constant, exactly like how the definition of the meter is based on the speed of light. How did scientists manage to do this?
How Is the New Kilogram Defined?
Short Version (TL;DR)
The new definition of the kilogram is based on the Planck constant. Since the Planck constant is a fundamental physical constant of nature, it will never change and neither will the value of the kilogram.
Long Version
To appreciate the whole story, we need to start back with Einstein and Planck.
One of Einstein’s crowning achievements is his famous equation E=mc2 which relates the mass of a particle to its energy content. A lesser known but still fundamental equation to modern science is the first “quantum” expression E=hv which relates the energy content of a wave to its frequency v and h, a value now known as the Planck constant, after Max Planck who first stated the expression.
Einstein’s equation shows that mass can be quantified in terms of energy, and Planck’s equation shows that energy can be understood in terms of a frequency v of some object (i.e. photons) times some integer multiple of the Planck constant h. This integer aspect is what makes Planck’s equation “quantum;’ energy comes in discrete packets called “quanta.”
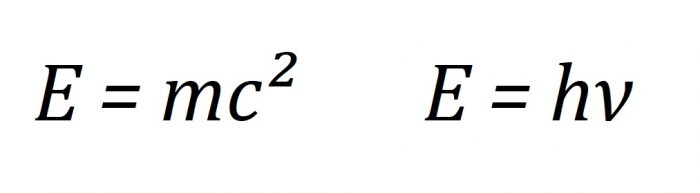
The two famous equations. Credit: Author
Combining the two equations tells us a surprising fact: mass is intrinsically related to the Planck constant h. The value of h has been measured to be 6.62607015 x 10-34 m2kg/s. The presence of kg in the units for the Planck constant is important because once the value of the Planck constant is defined, it means the kilogram is defined too.
Historically, the device used to measure the value of h is known as the Kibble balance, a device designed by British physicist Brian Kibble that measures mass precisely using electrical measurements. By the 1980s, Kibble balances using a defined mass were being used to measure the value of the Planck constant.

A Kibble balance at NIST laboratories in Gaithersburg, Maryland. Credit: Public Domain
In 1999 two physicists Peter Mohr and Barry Taylor published a letter proposing that a fixed value be assigned to the Planck constant to serve as the base for a new kilogram measurement. Mohr and Taylor reasoned that since exactly defined masses could be used to precisely measure the Planck constant, then defining the Planck constant should give us a way to precisely measure an unknown mass. Setting an exact value of h would let scientists work backward to calculate the exact value of 1 kilogram that makes the constant match up with the fixed value.
Mohr and Taylor’s proposal was endorsed in principle by the CGPM, on the condition that at least three experiments must make measurements of the Planck constant with an uncertainty of fewer than 50 parts per billion, an extremely tall order for the day. In 2017, the international scientific community finally succeeded in making these measurements, which kicked off the most recent redefinition of the metric system.
So finally, the new kilogram is defined by taking the fixed numerical value of the Planck constant h as 6.62607015 x 10-34 m2kg/s. So basically, whatever mass makes a Kibble balance spit out that value for the Planck constant is defined as exactly 1 kilogram. Instead of using a defined mass to measure the Planck constant, a fixed Planck constant is used to define the exact mass of a kilogram. The benefit is that the Planck constant is a universal constant and does not change, and so the value of the kilogram will never change. Notice that this new definition of the kilogram is dependent on the SI definitions of the meter and the second.
How Does This Redefinition Change Science?
That is the beautiful thing; the redefinition of the kilogram will not change scientific practice in the slightest! The new definition of the kilogram was exactly selected to sync up as well as possible with old definitions, so scientific instruments do not need to be recalibrated and nothing about measuring techniques will change. In fact, this shift signifies more of a reordering of our conceptual apparatus for doing science rather than a novel discovery in its own right.
The kilogram was not the only unit redefined either. The ampere (unit of electric current), Kelvin and mole were also redefined in a similar manner by taking fixed values of the elementary electric charge constant e, the Boltzmann k, and the Avogadro constant NA, respectively.
Even though it will have little to no effect on scientific practice, the redefinition of the kilogram completes the century-long search for a system of units that are based on fundamental aspects of reality itself. The idea is that the new definitions will remain the new standard for scientific measurement for all times and all peoples—a truly universal system of measurement that is based on nothing but nature herself.








