
Proteins are organic macromolecules that perform a multitude of function in the body. The monomers (individual units) of proteins are called amino acids. Most amino acids are composed of hydrogen (H), carbon (C), oxygen (O) and nitrogen (N). Aside from water, amino acids (in the form of proteins) makes up the majority of the bulk of human tissue. There are over 500 known amino acids in the human body which are classified with respect to polarity, shape, and attached functional groups. In the human body, amino acids with an amine and carboxyl groups are of particular importance. Proteins perform a staggering array of functions in the human body, so the amino acids that constitute them are necessary for life as we know it.
The conglomeration of amino acids into a single protein is the end product of gene expression. Instructions for creating the protein are encoded in the sequence of nucleotide bases contained in a single segment of DNA. RNA transcribes these instructions encoded in DNA in the form of mRNA. Strands of mRNA contain codons, 3-base segments, where each group of three bases corresponds with a particular amino acid. tRNA binds to the corresponding base segments, carrying along with it the specified amino acid. The amino acids are chained together to form a polypeptide stand. Several polypeptide strands chained together to make a full-blown protein.
Aside from being the base units of proteins, amino acids themselves play important biological roles. Amino acids like glutamate and gamma-aminobutyric acid (GABA) regulate the firing of neurons in the brain, while others like proline and glycine are components of biological structures and substructures.
Amino Acids: The Basics

The basic structure of an amino acid. Credit: WikiCommons CC0 1.0
Amino acids have a common structure that consists of a central carbon atom bonded to an amino group (NH2) and a carboxyl group (COOH). In vivo, amino acids typically exist in an acidic solution, where they have been deprotonated, each group losing hydrogen, and so have a positive charge.
The “R” stands for a bonded side chain, which is the part that is unique to each kind of amino acid. The particular R chain determines the identity of the amino acid. If the R group is hydrogen, then it is glycine; if it is a benzene ring then it is phenylalanine. The properties of the side chain determine many of the physical properties of the whole amino acid, such as its solubility, acidity, polarity, and whether it is an oxidizer or a reducer.
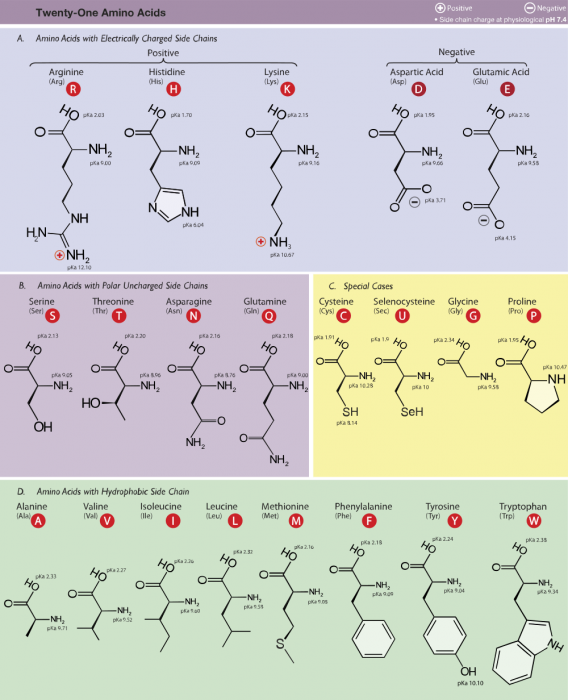
Credit: “Amino acids” Dancojocan via WikiCommons CC BY-SA 3.0
Even though there are over 500 known amino acids, human DNA only codes for about 20. These 20 amino acids are the ones that actually make proteins. Various other amino acids are synthesized and have uses outside of making proteins.
Of these 20 amino acids, the human body is unable to synthesize 9 using existing compounds in the body. These 9 “essential” amino acids must be consumed from external sources. The 9 essential amino acids are phenylalanine, valine, threonine, tryptophan, methionine, leucine, isoleucine, lysine, and histidine
How Do Amino Acids Make Proteins?
Proteins are chains of polypeptides, stands made of linked amino acids. Amino acids are linked to neighboring amino acids via covalent bonds called peptide bonds. Peptide bonds occur when the carboxyl group of one amino acid bonds with the amine group of another. The reaction removes a single molecule of water and results in a covalent bond between carbon and nitrogen.
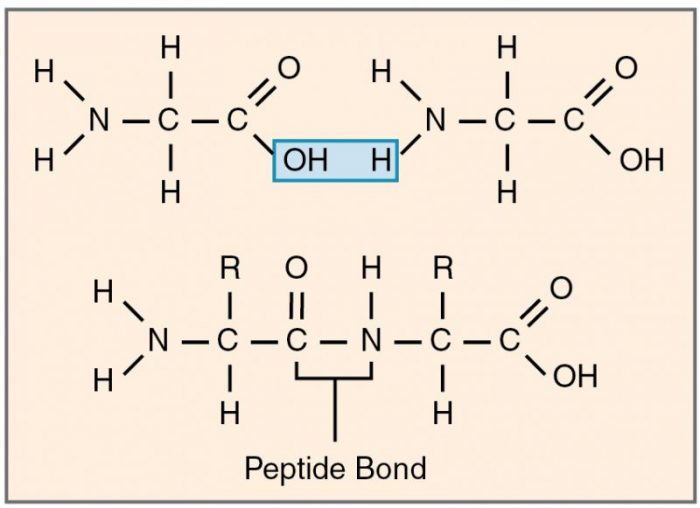
A peptide bond is a dehydration reaction that removes a molecule of water. Credit: “Peptide Bond” Openstax via WikiCommons CC BY-SA 3.0
Since amino acids are geometrically asymmetric, polypeptide chains have a direction. One end terminates in a nitrogen base amine group, while the other ends in a carbon-based carboxyl group.
Amino acids are physically assembled into polypeptide strands during the process of RNA translation. During translation, a transcribed mRNA bonds to a ribosome. Other RNA molecules, called tRNA, attach themselves to the mRNA via complementary nucleotide base-pairing. Molecules of tRNA carry the amino acids that are specified by the mRNA codons. For example, if the mRNA codon is ‘AUG’ then the corresponding tRNA base pairing is ‘TAC’, which specifies the amino acid methionine. Likewise, the codon UGG encodes for the amino acid tryptophan. tRNA bonds to mRNA placing the amino acids next to each other, which then form peptide bonds and create a polypeptide chain
Generally, after construction, the polypeptide chain undergoes a few post-translation modifications, where groups may be added or certain sequences are modified slightly to other sequences.
Structure Of Proteins
Each protein is uniquely specified by the sequence of amino acids it contains. The order of these amino acids affects the functioning of the protein. Changes in DNA can result is a change in the expressed sequence of amino acids, which will affect the functioning of the associated protein. For example, the symptoms associated with sickle cell anemia are due to one difference in the amino acid sequence that specifies hemoglobin. The glutamic acid that takes up the 6th spot is replaced by valine. The result is that created hemoglobin proteins have a long thread-like shape. This thread like shape changes the shape of blood cells, causing them to stick to cell walls and impair blood flow.
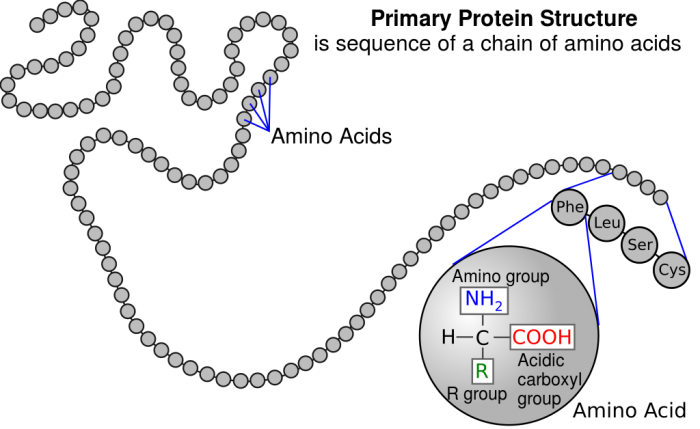
Proteins are formed of long chains of peptides, which are made of amino acids. Credit: WikiCommons CC0 1.0
In addition to the sequence of amino acids, dubbed the primary structure, proteins have a secondary structure that refers to the geometric folding of the peptide chain backbone. There are two main kinds of secondary structures seen in the human body, called an α-helix and a β-pleated sheet.
In an α-helix structure, the peptide chain twists around itself like a coiled snake. The carbonyl group on one amino acid is hydrogen bonded to the amine group 4 spots down. The resulting structure is a tightly coiled helix that is held together by hydrogen bonds. β-pleated sheets, on the other hand, consists of linear chains of polypeptides stacked laterally, held together by hydrogen bonds between the amine and carbonyl groups of neighboring amino acids. Some amino acids are more likely to form α-helix structures than β-pleated sheets and vice versa.
Lastly, proteins have an overall 3-D structure called its tertiary structure. The tertiary structure is the three-dimensional geometry of the entire protein and is determined by the interaction of the R side chains. R chains interact through virtually every known non-covalent bond. Whether or not R groups are hydrophobic or hydrophilic affects the orientation of the protein.
Many proteins consist of just a single polypeptide chain, but some are made of multiple polypeptide chains conglomerated together. These kinds of polypeptide chain proteins have a quaternary structure, one that is determined by the interactions of the respective tertiary structures. Hemoglobin is one such protein that is composed of 4 polypeptide chains 2 each of the α and β structures.
Proteins require certain pH values and temperatures to retain their shape. When temperatures of pH values get too high or low, the intermolecular interactions holding the protein together might get disrupted and the protein will unravel, losing its shape. unraveled proteins are called “denatured” proteins, and no longer can perform their functions. For example, very high fevers are dangerous because the heat can denature proteins in the brain, unraveling and preventing them from functioning.
Other Uses Of Amino Acids
Other amino acids that do not construct proteins play important biological roles. One striking example is gamma-aminobutyric acid (GABA). GABA is one amino acid that is crucial for neurological functioning. GABA in the brain is the main neurotransmitter that regulates the firing of action potentials int he brains neurons. Specifically, GABA-mediated inhibition is a negative feedback process in the brain that dampens neurons, preventing them from firing. GABA bonds to structure in the axons, modulating the flow of potassium and sodium ions, which modulated the firing of action potentials. GABA is also responsible for regulating muscle tone in mammals.
Amino acids also serve as precursors to other important biological molecules, like serotonin, dopamine, epinephrine, and nucleotides.
To sum up, amino acids are the monomers of proteins. Amino acids consist of a central carbon atom attached to an amine and carboxyl group, bonded to a side chain. The nature of the bonded side chain determines the specific identity of the amino acid and its bonding behavior.
During the process of RNA translation, sequences of amino acids are pieced together based on the information encoded in the relevant section of DNA. Amino acids form a covalent bond between their amine and a carboxyl group, forming long chains called polypeptide strands. The polypeptide strands will then fold into the appropriate structure, forming a fully fledged protein. In some cases, multiple polypeptide strands link together to create proteins that are complex of multiple polypeptide strands. The exact identity of the protein and its associated functioning is determined by its exact sequence of amino acids. A change in that sequence can bring about a change in the functioning of the protein. Proteins that lose their shape are denatured and normally are no longer functional.









