Glucose (C6H12O6) is an organic macromolecule that is essential for the metabolism of essentially all eukaryotic organisms. Glucose is a monosaccharide (simple sugar) and is the most abundant carbohydrate. Glucose is normally stored in the body in the form of starch or glycogen. Glucose provides the raw materials needed for cellular respiration and the production of ATP.
“The brain’s preferred source of fuel is glucose/carbohydrates. And when you go on a low-carb/high-protein diet, your brain is using low-octane fuel. You’ll be a little groggy, a little grumpy.” — Jack LaLanne
The molar mass of glucose can be calculated by multiplying the molar masses of its atomic constituents by their frequency in a single molecule and adding those values together. Glucose is composed of hydrogen (H), carbon (C), and oxygen (O) The molar mass of H is 1.0079, the molar mass of C is 12.0107, and the molar mass of O is 15.9994. In one molecule of glucose, there are 12 hydrogen, 6 carbon, and 6 oxygen atoms. So, altogether, the molar mass of a single molecule of glucose is equal to:
1.0079(12)+12.0107(6)+15.9994(6) = 180.16 g/mol
Glucose has a molar mass of 180.16 g/mol. One mole of glucose molecule has a mass of 180.16 g.
Molar Mass
The molar mass of a given substance is a quantitative measure that tells you the mass of 1 mole of that substance. In chemistry, molar mass is understood as a physical property that is defined as the mass of a substance divided by the amount of that substance.
The measure of molar mass (g/mol) is based on the SI unit for quantity, the mole (not to be confused with the cute burrowing mammal). 1 mole is defined as an amount of substance that contains exactly 6.0221476 × 1023 constituent particles. Just like the words “million” and “billion,” the word “mole” stands for a specific quantity of things; approximately 602,214,150,000,000,000,000,000 of them. If I had one mole of apples, I would have 602,214,150,000,000,000,000,000 apples, if I had one mole of hydrogen atoms, I would have 602,214,150,000,000,000,000,000 of them.

A mole with 1 mole. Credit: “Mole” Andi via Flickr CC BY-SA 2.0
Every element has a molar mass, i.e. a measure of how much mass one mole of that element has. The molar mass of any element can be determined by multiplying that elements standard atomic weight (listed on the periodic table) by the molar mass constant Mu=1g/mol. Hydrogen, for instance, has a standard atomic weight of 1.00794. To find the molar mass of hydrogen, we simply multiply this number by the molar mass constant to get 1.00794 g/mol. So, hydrogen has a molar mass of 1.00794 g/mol; that is, 6.0221476 × 1023 hydrogen atoms would together weigh 1.00794 grams.
To find the molar mass of a molecule or an ionic compound, all one has to do is first multiply the molar masses of the constituent elements by their frequency in the compound, and add the total values together. One can determine the relative atomic frequencies of a composition by the compound’s molecular formula. In other words, the molar mass of a compound is equal to the sum of the molar masses of its constituent atoms.
“The production and consumption of glucose, and hence, the blood sugar level, are controlled by a functional endocrine equilibrium.” — Bernardo Houssay
For example, water is made of 2 hydrogen atoms and 1 oxygen atom and has a molecular formula of H2O. To find the molar mass of water, one first needs to find the molar mass of hydrogen and oxygen, multiply those values by their relative frequency in a single molecule of the compound, and add the totals together. Hydrogen has a molar mass of 1.00794 and oxygen has a molar mass of 15.9994. Each molecule of water has 2 hydrogen atoms and 1 oxygen atom, so the molar mass of water equals:
1.00794(2) + 15.9994(1) ≈ 18.02 g/mol
So one mole of water molecules would have a weight of 18.02 grams.
Importance Of Molar Mass
Molar masses are important because they figure in equations used to predict the physical and chemical behavior of substances. Most importantly, the concept of molar mass serves as the bridge between mass and amount of substance because it is generally impossible to directly count how many particles are in a substance. We can measure mass though, so knowing the molar mass allows us to indirectly measure the number of particles in a substance by measuring its mass.
Experimental setups often reference moles and molar masses in their steps. Say an experiment calls for 3 moles of water. We cannot directly count individual molecules of water (it would take way too long even if we could) so instead we can rely on the molar mass of water to figure out how much water we need. 1 mole of water has a mass of 18.02 grams, so if an experiment calls for 3 moles of water, we know that we need 18.02(3) = 54.06 grams of water. Likewise, if an experiment called for 0.7 moles of carbon, we know that we need 12.0107(0.7) = 8.407 grams of carbon.
Molar Mass Vs Molecular Mass
It is important to not confuse the concepts of molar mass and molecular mass. The molar mass of a compound tells you how much one mole of a substance weighs but it does not really tell you anything about the weights of the individual molecules. The measure of the mass of an individual molecule of a compound is its molecular mass. Molecular masses are measured in daltons (Da), named after the father of atomic theory, John Dalton. Molecules of the same compound can have different molecular masses because they can be composed of different isotopes of the same element. Water may have a molar mass of 18.02 g/mol, but individual water molecules can have a weight that ranges from 18.011 Da to 22.028 Da, due to the presence of different isotopes of hydrogen and oxygen. The molar mass can then be seen as a measure of the average molecular masses of the individual molecules in one mole of a substance.
Molar Mass Of Glucose
Using the above definitions, we can determine the molar mass of glucose step by step. First, we look at the molecular formula to determine the atomic constituents and their relative frequencies in a single molecule. glucose has a molecular formula of C6H12O6, so a single molecule of glucose contains 6 carbon atoms, 12 hydrogen atoms, and 6 oxygen atoms.
The molar masses of carbon, hydrogen, and oxygen are 12.0107 g/mol, 1.00794 g/mol, and 15.9994 g/mol, respectively. These values can be determined by multiplying the standard atomic weight for each element by the molar mass constant. Next, we can multiply these values by the frequency of each element, so:
12.0107 × 6
1.00794 × 12
15.9994 × 6
Adding all these values together will give us the total molar mass of glucose:
1.0079(12)+12.0107(6)+15.9994(6) = 180.16 g/mol
Glucose As A Compound
Glucose is a simple sugar (monosaccharide) that is ubiquitous in living organisms. It is the main source of metabolic energy in virtually all living creatures and is physically abundant in many structures in the body. Glucose is classified as a hexose (six carbon atoms) and has several distinct polymorphs. The most common and naturally occurring form, D-glucose, consists of a cyclical chain of 5 carbon atoms each bonded to a hydrogen and hydroxyl group, closed off with a carbon-containing aldehyde group (R[CHO]). In certain solutions, glucose will unravel from its cyclical arrangement to form a linear chain of carbon atoms capped with the aldehyde group.
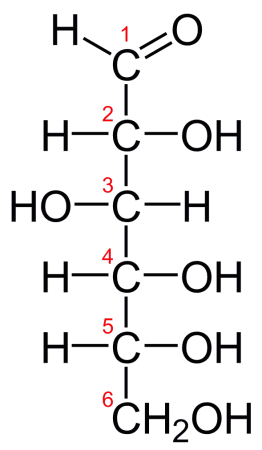
Fischer projection of D-glucose. Credit: “D-glucose chain” via WikiCommons CC0 1.0
All kinds of glucose are colorless and are easily dissolved in water, alcohol, and other organic solvents. Its solubility makes it an essential compound for biological processes. Photoautotrophs, like plants, produce their own source of glucose via photosynthesis, but heterotrophs, like humans and all other mammals, must get their glucose from external sources. Glucose is the main ingredient that gets processed during cellular respiration.
During cellular respiration, one glucose molecule is broken down into two pyruvate molecules in a process called glycolysis. The pyruvate molecules are then converted into acetyl-CoA, which is processed according to the Krebs cycle. The energy produced during the Krebs cycle is the main driver of oxidative phosphorylation, the process by which the body actually produces ATP, the fundamental energy currency of biochemical processes. ATP drives literally every biological reaction in the body, so without a steady supply of glucose, the body will not be able to produce its fuel. For every one molecule of glucose, one full turn of the cellular respiration cycle has a theoretical yield of 38 molecules of ATP. In practice, inefficiencies in chemical reactions or loss of energy during oxidative phosphorylation give an actual yield of about 33-34 molecules of ATP per molecule of glucose.
Glucose in the blood is called blood sugar. Normal bodily functioning requires some level of blood sugar, but too much can be harmful. Elevated levels of blood sugar, called hyperglycemia, can lead to nausea, fatigue, stomach pains, blurred vision, and frequent urination. Diabetics lack the ability to produce insulin, the hormone that regulated blood sugar levels, so diabetics are at risk for hyperglycemia. In severe cases, high levels of blood sugar can restrict oxygen flow through the capillaries, resulting in infection and tissue death.
To recap, every element has a molar mass, a measure of how much one mole of that substance weighs. The molar mass of an element can be determined by multiplying the standard atomic weight by the molar mass constant g/mol. The molar mass of a compound is equal to the sum of the molar masses of its constituent elements. The molar mass of a compound can be determined by multiplying the molar masses of the individual elements by their relative frequency in a molecule of a compound and summing the total values. In the case of glucose (C6H12O6), glucose has a molar mass of 180.16 g/mol.









