
Glucose, glutamine, and fatty acids, the major fuel for mammalian cells, are taken up not only to produce the necessary energy but also to produce all the biomolecules required to build new cells after division. Nutrient uptake is achieved through specific transporters. The role of these transporters in cell survival under nutrient restriction in certain areas of a tumor mass is not clear and it was the main interest of our work.
Cancer cells divide further and faster than normal cells do, so they have to choose between either oxidizing nutrients to get energy or synthesizing new biomass, generally in favor of the second in order to obtain the necessary building blocks to create new cells. This phenomenon implies that glucose does not finish its oxidative circuit into the mitochondria to produce ATP. Instead, cancer cells reprogram their metabolism through a mechanism that only recently has begun to be understood.
First proposed by Otto Warburg in the 20s, in the last decade, potential changes of cancer cell metabolism have focused the attention of many researchers worldwide. Therefore, cancer cells have become “addicted” to both glucose and glutamine, but to internalize them at higher rates, they have to increase their transporter production and subsequent exposure at the plasma membrane.
In addition to a reduced O2 microenvironment, restriction of glucose supply is one of the boundaries that cancer cells have to scope to live inside tumors. Strategies to access to nutrients and oxygen are frequently developed by cancer cells. The increase of blood vessels biogenesis is a well-known mechanism to battle those limitations. For many years, the attention of researchers and pharmaceutical companies have been focused on the development of drugs that limit blood vessels supply in tumors. Moreover, at the cellular level, cancer cells follow three major strategies to manage the situation facing nutrient limitations. Usually, cells either increase the acquisition of nutrients by upregulating glucose or glutamine transporters, reprogram metabolic routes to create new biomass, and/or finally increase their ability to obtain metabolites and nutrients from the microenvironment to balance the limiting access to nutrients.
In fact, the increasing uptake of glucose by cancer cells is the base of positron emission tomography (PET) which is employed in clinical practice for the diagnosis, the staging, as well as the screening of their response to treatments.
Considering the above, glucose deprivation appears to be a logic approach to induce the death of cancer cells. However, several cancer cell types develop new adaptative responses to survive even under low concentration of nutrients. Our group and others have demonstrated the important role of glucose transporters and glucose reprogramming metabolism in the progression and survival of prostate cancer cells. The general purpose of this work was to explore the mechanism that favors prostate cancer survival after glucose removal and in particular, we look for biological differences between androgen sensitive and insensitive prostate cancer cells.
To this aim, glucose was removed from culture media of either androgen-sensitive (early stage) and androgen-insensitive (late stage) prostate cancer cells and their response, in terms of growth and survival, was approached in this study.
Androgen-sensitive prostate cancer cells are more resistant to glucose deprivation
First, although androgen-insensitive prostate cancer cells are a cell model for an advanced stage of prostate cancer, a hormone resistant phenotype, interestingly, they showed a higher sensitivity to glucose removal than androgen-sensitive prostate cancer cells.
In addition, we observed that in androgen-sensitive cells, glucose removal promoted an increase in the levels of its own transporter (GLUT1). After glucose removal, we found higher levels of free radicals inside cells and an activation of androgen signalling that seemed to be linked. In fact, we observed that increasing the levels of hydrogen peroxide (H2O2) activates androgen signalling in prostate cancer cells.
Overproduction of glucose transporters protect prostate cancer cells from oxidative cell death
In the same way, we found that ectopic overexpression of the levels of glucose transporter type 1 (GLUT1) significantly reduced the levels of cell death after glucose removal. Since glucose is removed from culture media, by increasing glucose transporters, we did not increase the uptake of glucose, and another unknown mechanism must be playing a role.
In fact, we found that overexpression of GLUT1 reduces the oxidative stress induced by glucose deprivation, the main function responsible for cell death in our model. This was confirmed because when cells were grown in the presence of antioxidants such N-acetyl cysteine (NAC), they do not die at all after glucose removal.
Therefore, we found that after glucose removal, levels of oxidative stress were enhanced. However, androgen-sensitive prostate cancer cells are able to survive because they activate androgen-dependent signaling pathways which subsequently lead to the expression of glucose transporters that play an antioxidant role in these cells. As mentioned above, this survival route is shut down in androgen-insensitive cells, thus explaining why they rapidly die after glucose removal from culture media.
GLUT1 glucose transporter acts as an oncoprotein promoting survival against nutrient deprivation in prostate cancer cells
As a conclusion, GLUT1 glucose transporter acts as an oncoprotein in several tumors. However, it does not only promote proliferation because of increasing glucose uptake in prostate cancer cells but, additionally, it has a significant protective role against nutrients deprivation. Glucose starvation rises oxidative stress that, in turn, activates androgen signaling and, as a consequence, GLUT1 production (Summary Figure).

Image published with permission from Rosa M. Sainz, Juan C. Mayo & Pedro Gonzalez-Menendez.
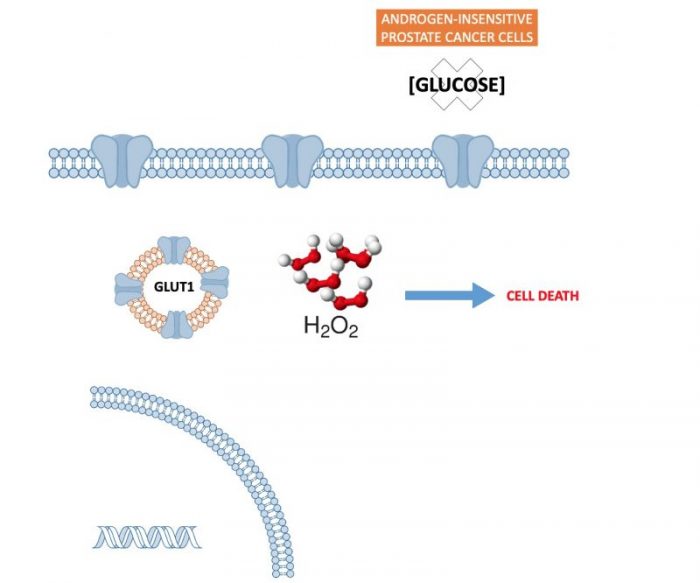
Image published with permission from Rosa M. Sainz, Juan C. Mayo & Pedro Gonzalez-Menendez.
After GLUT1 production and exposure in cell membranes, a rise in endogenous antioxidants protects prostate cancer cells from cell death. In prostate cancer, glucose transporters are usually augmented in aggressive tumors so that these results support that in addition, they might also protect cancer cells from a low glucose microenvironment.
These findings are described in the article entitled GLUT1 protects prostate cancer cells from glucose deprivation-induced oxidative stress, recently published in the journal Redox Biology. A team including Rosa M. Sainz, Juan C. Mayo and Pedro Gonzalez-Menendez from the University of Oviedo and the “Institut de Génetique Moléculaire Centre National de la Recherche Scientifique,” University of Montpellier, conducted this work.









