
Ebola virus and Marburg virus (MARV) are closely related filoviruses that cause severe hemorrhagic fever disease in humans and nonhuman primates and have high fatality rates.
Sexual transmission of filoviruses was first reported in 1968 when MARV was identified in Germany as the first recorded filovirus. Several smaller outbreaks of filovirus disease have occurred since then, but the 2013–2016 Ebola virus disease outbreak in Western Africa was the largest in recorded history. A total of 28,646 cases and 11,323 deaths in 10 countries worldwide have been reported by the World Health Organization.
Ebola virus genomic RNA has repeatedly been detected in semen of Ebola virus disease survivors up to 18 months after recovery, and sexual transmission of the virus has been implicated in the initiation of entirely new Ebola virus transmission chains during the outbreak in Western Africa. However, the mechanism that led to testicular filovirus persistence and filovirus shedding into semen remains elusive.
The large cohort of outbreak survivors — some 17,323 individuals — has quickly created a new research focus in the study of filoviruses. Scientists are now working to understand how persistent filovirus infection can linger in the immune-privileged organs — including testes, eyes, and brain tissue — of survivors who have cleared the virus from their bloodstreams and demonstrate no clinical signs of disease.
In our 2018 article published in Cell Host & Microbe, we discovered that persistent MARV infection of seminiferous tubules (an immune-privileged site that harbors sperm production) is a relatively common event in cynomolgus macaques that survived infection after antiviral treatment. This paper elucidates the primary processes leading to testicular persistence and sexual transmission of filoviruses and lays the foundation to develop therapeutics to clear testicular viral persistence and ultimately prevent sexual transmission of these viruses.
MARV Persists in the Seminiferous Tubules of Testes of Cynomolgus Macaque Survivors
The mammalian testes consist of two main compartments: seminiferous tubules and interstitial tissue. The interstitial tissue is mainly comprised of testosterone-producing Leydig cells, in addition to the vascular structures. The seminiferous tubules are the site of the germination, maturation, and transportation of sperm cells within the testes, which possess a special immunological environment — specifically, they protect sperm cells from the body’s immune attack by effectively blocking antibodies and lymphoid cells from entering and preventing invading microbial pathogens.
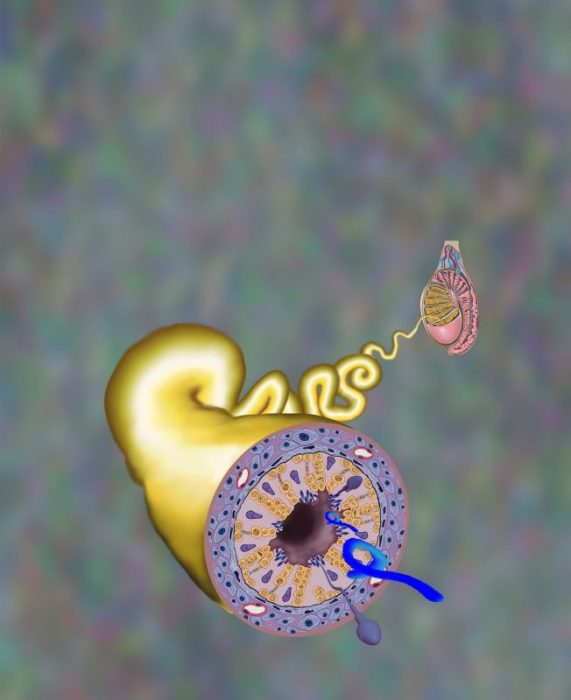
llustration of a macaque testis. Marburg virus (blue) hides in the coiled tubules known as the seminiferous tubules, sites of sperm production and immune privilege. Art by William F. Discher, USAMRIID
Using a highly sensitive and specific molecular detection assay called in situ hybridization, we discovered that 22 of 73 (30.1%) monkeys that survived MARV infection after antiviral treatment had persistent infection in the seminiferous tubules of the testes. Interestingly, MARV was not detected in common target organs, such as the liver, spleen, and lymph nodes, in survivors with testicular MARV persistence.
Severe Damage and Inflammatory Cell Infiltration in Testicular Sites with MARV Persistence
By using immunofluorescence staining and confocal imaging, we determined that Sertoli cells, which nourish and protect developing sperm cells, are the main cellular reservoir of MARV persistence in the seminiferous tubules. Our molecular pathology studies further revealed that severe tissue damage, including seminiferous tubule degeneration, spermatogenic cell depletion, breakdown of the blood-testis barrier, and inflammatory cell infiltration, were observed in the sites of MARV persistence. Additionally, local infiltration of immunosuppressive CD4+Foxp3+ regulatory T cells were detected in the sites of persistent MARV infection. These immunosuppressive regulatory T cells may sustain viral persistence.
Notably, despite persistent MARV infection, the overall reproductive function of the testes appears to be unaffected. Normal spermatogenesis and tissue morphology were found in the majority of testes tissue with focal MARV infection. This observation could explain why human Ebola virus disease survivors shed viral RNA in semen for extended periods of time in the absence of clinical symptoms.
Early Administration of Therapeutics Prevents MARV Persistence in Testes
Our study revealed that it takes significantly longer for MARV to infect the testes than common target organs such as liver, spleen, and lymph nodes. Our data also suggest that, in order to prevent persistent filovirus infection in the testes, patients should take therapeutic interventions as early as possible to clear infection before the virus starts to infect the testes. Indeed, our therapeutics study demonstrated that early therapeutic intervention can efficiently prevent testicular MARV persistence. These findings have important implications for the ongoing outbreak of Ebola virus disease in the Democratic Republic of the Congo.
These findings are described in the article entitled Persistent Marburg Virus Infection in the Testes of Nonhuman Primate Survivors, recently published in the journal Cell Host & Microbe. This work was completed by Kayla Coffin, Jun Liu, Travis Warren, Candace Blancett, Kathleen Kuehl, Donald Nichols, Jeremy Bearss, Christopher Schellhase, Cary Retterer, Jessica Weidner, Sheli Radoshitzky, Jennifer Brannan, Anthony Cardile, John Dye, Gustavo Palacios, Mei Sun, Sina Bavari, and Xiankun Zeng from the United States Army Medical Research Institute of Infectious Diseases, as well as Jens Kuhn from the Integrated Research Facility at Fort Detrick, National Institute of Allergy and Infectious Diseases, National Institutes of Health.









