
A heterogeneous mixture is a combination of two or more substances, creating a new substance that has nonuniform composition. Within the new substance, the concentration of the constituent substances will vary between regions of the substance, so it is possible for one of the chemicals to be more prominent towards the top of a heterogeneous mixture than at the bottom. You can typically see the two separate components in a heterogeneous mixture.
Examples of heterogeneous mixtures include things like soda, concrete, soil, cereal and milk, and orange juice.
Mixtures: Heterogenous And Homogenous
The term mixture is applied to two or more substances combined together, creating a compound combination where the different parts of the mixture have their own separate chemical identity. Heterogeneous mixtures are mixtures which are made up of different substances mixed together. In a mixture, the chemical bonds that exist between the different components of the mixture are exchanged. New bonds are not established nor are bonds broken. While the chemical properties may not change as a result of combining two or more components, the newly combined substance may have additional physical properties that were not characteristic of these constituent parts. As an example, the boiling point of a water-alcohol mixture is higher than pure alcohol and lower than pure water.
A heterogeneous mixture is a mixture which has separately identifiable substances composing it, it has a non-uniform composition. Different parts of the composition will have higher or lower concentrations of the individual substances that comprise the mixture, and in a heterogeneous mixture, there will be at least two substances possessing different phases distinct from each other. In contrast to the nonuniform composition of heterogeneous mixtures, homogeneous mixtures are in a state of equilibrium where they possess the same composition and phase, regardless of where in the composition the sample is taken.
The difference between heterogeneous and homogenous mixtures is often one of scale. As an example, a seemingly homogeneous air sample can appear as heterogeneous when examining the sample at the level of molecules. It is also possible for a heterogeneous mixture to be made out of a single element. As an example, carbon can be in different states such as diamonds and pencil lead, but a mixture of the two would be heterogeneous.
Note that the definition of a heterogeneous mixture is somewhat different in the materials sciences and in chemistry, where a heterogeneous mixture is defined as a mixture which contains components existing in different phases.
Just because two chemicals are mixed together doesn’t mean that a mixture will always result. Sometimes a chemical reaction occurs when two chemicals are mixed together and as a result, the reactants change their identities. For instance, baking soda and vinegar combined together produce water and carbon dioxide, and therefore a mixture does not result. Combining bases and acids also don’t produce mixtures.
Terms To Describe Mixtures
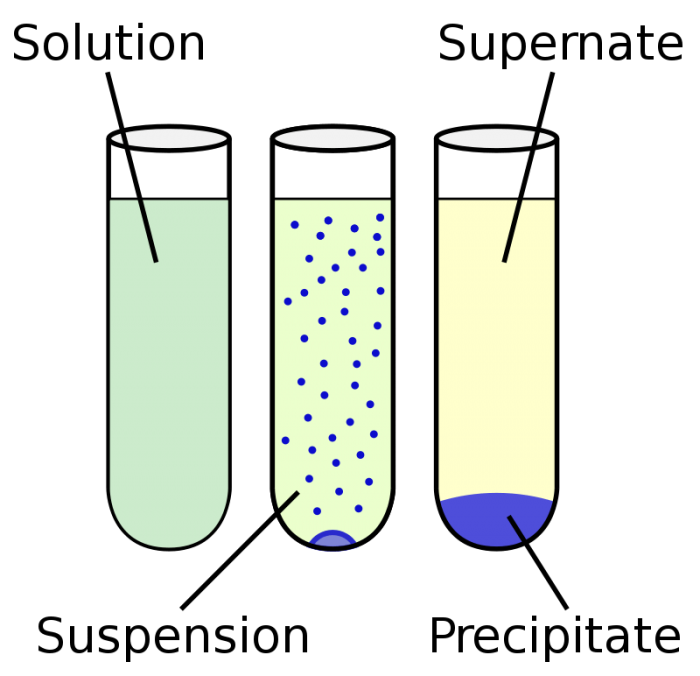
Solutions with identifiable particles are heterogenous solutions. Photo: By Vectorized by ZooFari; raster by ZabMilenko – Own work based on: Chemical precipitation diagram.png, Public Domain, https://commons.wikimedia.org/w/index.php?curid=9733498
It is possible to describe mixtures with terms that refer to the particle size of the mixture’s components. The term solution is usually applied to mixtures with extremely small particle sizes, with the particles typically being less than 1 nm in diameter. Solutions are typically stable, and the different chemical components cannot be separated from one another by the processes of centrifuging or decanting. Examples include oxygen dissolved into water, air, and gelatin.
A colloid is the term given to solutions which look homogeneous when examined with the naked eye but when examined under a microscope the separation of the particles can easily be seen. Colloids are physically stable much like solutions, but unlike solutions, the size of particles usually ranges between 1 nm and 1 µm. Colloids are known for exhibiting the Tyndall effect, which refers to the scattering of light as it bounces off particles in a fine suspension or colloid. Much like solutions, the components of colloids can’t be separated through the process of decantation, yet the components of the colloid can be isolated through centrifugation. Examples of colloids include smoke, blood, whipped cream, and hairspray.
Suspensions are mixtures where the particles are large enough that the mixture appears heterogeneous even to the naked eye. Typically a suspension requires the use of a stabilizing agent to keep the particles from separating. Suspensions also display the Tyndale effect, much like colloids. The constituent parts of suspensions can be isolated/separated from one another through centrifugation or decantation. Examples of solutions include things like mud, dust suspended in the air, granite, vinaigrette, and sand.
Homogenization
The process of homogenization is what turns a heterogeneous mixture into a homogeneous mixture. One of the most notable examples of homogenization is the process that produces homogenized milk. Natural milk separates into different layers, with the liquid on the bottom and a layer of cream on top, typically only appearing as a homogenous mixture when shaken. By contrast, homogenized milk is processed to ensure that the components of the milk are stable and don’t separate out the way natural milk does.
Examples Of Heterogeneous Mixtures

Photo: Myriams-Fotos via Pixabay, CC0
Let’s take a look at some examples of heterogeneous mixtures. Soda is one example of a heterogeneous mixture, being comprised of carbon dioxide, sugar, and water. The carbonation in the drink is what creates the bubbles. The water, sugar, and any flavorings in the soda comprise a chemical solution, but the carbon dioxide appears rather randomly throughout the solution, and this lack of uniform distribution makes soda a heterogeneous mixture. Concrete is another heterogeneous mixture with water as an ingredient, being made out of water, cement, and an aggregate. Soil is a heterogeneous mixture because it is comprised of various minerals and elements arrayed in a nonuniform fashion. Soil frequently has clumps with higher concentrations of a mineral than the area immediately surrounding that clump of soil. Cereal in the milk is an example of a heterogeneous mixture because not only is the serial separate from the milk, but milk itself can potentially be heterogeneous as discussed above. Orange juice with pulp is another example of a heterogeneous mixture, with the pulp consisting of a different state (a solid), suspended in the surrounding liquid.
Homogeneous Mixture Examples
As previously stated, homogenous mixtures are mixtures which are consistent/uniform throughout the entire mixture. Homogenous mixtures can also be referred to as solutions. Let’s go over some examples of homogeneous mixtures. Homogenous mixtures are those like blood plasma, coffee, vinegar, and laundry detergent.
Blood plasma is a colorless fluid that contains cells, found within the blood, suspending them in a fluid. This fluid accounts for about half of the blood’s volume. A cup of coffee can also be considered a homogenous mixture, as typically the grounds have been strained out of the coffee when it is brewed, leaving a fluid where the water and coffee extract has been combined together into a uniform mixture. If sugar or cream is added into the coffee it becomes a heterogeneous mixture until the substance has been uniformly distributed and the coffee is a homogeneous mixture once more. Vinegar is a homogeneous mixture comprised of some large percentage of acetic acid and other ingredient chemicals, perhaps flavorings. While there are multiple components of the vinegar, these chemical components are uniformly distributed and therefore the mixture is homogeneous. Laundry detergent is a mixture made out of various kinds of soaps/cleaning chemicals, but when you pour the detergent into a washing machine these detergents are homogenous and you are unable to distinguish the individual cleaning chemicals.
Getting An Intuition For Mixtures

The different colors of these candies means that pulling a random sample would likely get you different colors, hence it is a heterogeneous solution. Photo: eismannhans via Pixabay, CC0
When you are trying to determine if a mixture is homogenous or heterogeneous in nature, it is important to keep a few things in mind. Remember that the components in a homogenous mixture will be present in the same proportion, so it doesn’t matter where in the mixture a sample is taken. Heterogeneous mixtures do not have this property and the composition of a sample is affected by where in the mixture the sample is taken. If you imagine a jar of candies, and all of the candies are red, this is a homogenous mixture as you will only get red candies no matter where you scoop out a handful. By contrast, a jar made up of red, green and blue candies will be a heterogeneous mixture as you will get different combinations of colors depending on where you take a sample.
The heterogeneity or homogeneity of a substance is often dependent on the scale that substance is observed at. In general, substances are more likely to be homogenous when viewed on a small scale. By contrast, on a macroscopic scale, a mixture is more likely to be homogenous in nature.









