Proteins are a type of biological molecule that is composed primarily out of amino acids. Proteins perform a vast array of functions in the human body including:
- catalyze metabolic reactions
- synthesize and copy DNA
- transport molecules within the cell
- signaling paths in the body
- active in the immune system
- form physical structures of cells
Proteins are made out of polypeptide chains, which are chains of amino acids arranged in a specific sequence. The order of amino acids in the polypeptide chain determines the shape and function of the protein. The body constructs proteins according to information stored in DNA. The sequence of nucleotide bases in DNA represents a particular sequence of amino acids. During transcription and translation, this information is copied in the form of RNA and read by ribosomes to make the encoded protein.
All in all, human DNA encodes for 21 different amino acids, which leads to a large pool of possible proteins. Human DNA has about 20,000 genes that encode for functional proteins. The exact number of different kinds of proteins in the human body is not known, though speculations have put the number from 100,000 to 1 million. Humans also require 9 amino acids that the body cannot synthesize, so we rely on external sources of protein in our food to satisfy this requirement.
10 Examples Of Proteins
10. Hemoglobin
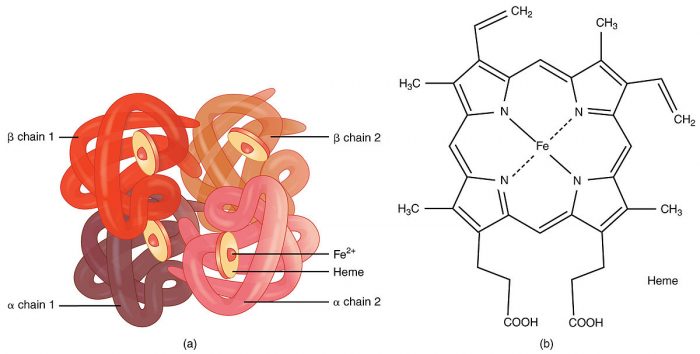
Credit: OpenStax via WikiCommons CC-BY 3.0
Hemoglobin is a metalloprotein located in red blood cells of all vertebrates and most invertebrates. Hemoglobin’s primary function in the blood is to carry oxygen from the lungs or gill to the body’s tissues. It also binds to and transports carbon dioxide. In mammals, hemoglobin makes up about 35% of the total volume of the cell. The presence of hemoglobin increase the oxygen content of the blood by 70 times compared to simply dissolving oxygen in the blood.
Hemoglobin contains iron which is why it can transport oxygen so effectively. There are several medical conditions characterized by a deficiency in hemoglobin. Sickle cell disease results from a mutation that causes an error in the construction of hemoglobin chains. The error causes red blood cells to flatten and take on a sickle-like shape. Anemia is also associated with lowered levels of hemoglobin.
9. Keratin
Keratin is a fibrous protein that is the main structural component of hair, nails, hooves, horns, claws, etc. Keratin is also found in epithelial cells and is what gives skin its mechanical strength and makes it waterproof. There are two major types of keratin. α-keratin is found in all mammals and makes up the hair and other external appendages. β-keratins are tougher and are mostly found in reptiles and birds, in the form of scales, beaks, claws, feathers, and shells. Keratins are also found in silk threads produced by insects and spiders.
Human DNA has 54 distinct genes that code for keratin which are located on chromosomes 12 and 17. Living cells can become “keratinized” in which the cells fill with keratin and loses their organelles and nucleus. The outermost layer of human skin is keratinized tissue.
8. Insulin
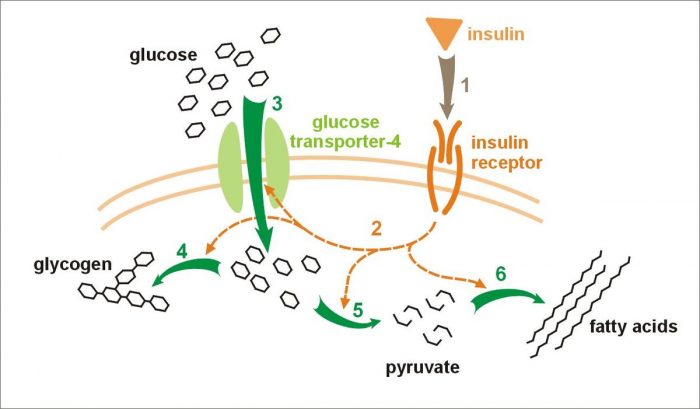
Credit: Meiquer via WikiCommons CC-BY 3.0
Insulin is a protein that is made primarily in the pancreas. The cells in the pancreas that synthesize insulin are called beta cells. The main function of insulin is to control the breakdown of glucose and regulate blood sugar levels. When blood sugar levels are high, beta cells in the pancreas secrete insulin which increases the rate of glycolysis and glycogenesis. When blood sugar levels are low, insulin production is inhibited.
Human insulin consists of 51 amino acids and has a chemical formula of C257H383N65O77S6. Several conditions exist that are characterized by insulin deficiency of insulin immunity. In patients with type I diabetes, the bodies immune system attacks its own beta cells making it so the pancreas produces little to no insulin. Type II diabetes results from an acquired immunity to insulin and subsequent decreased insulin production. Symptoms for both conditions include high blood sugar, low insulin levels, frequent urination, and increased thirst.
7. Myoglobin
Myoglobin is a structural protein found primarily in muscle tissue of vertebrates. Myoglobin is related to hemoglobin and serves a similar purpose—it provides a site in the muscles for oxygen to bond to. Myoglobin is the substance responsible for the distinct color of red meat. The more saturated with oxygen myoglobin is, the redder the tissue. Myoglobin is present in the blood only after acute muscle injury, which makes it a potential sign of injury or illness.
Although it is a well-studied protein, the exact function of myoglobin is not conclusively known. Myoglobin seems to function to give extra storage space for oxygen in the muscles, which is useful for organisms that need to hold their breath for a long time. For example, whales and seals have muscles with very high levels of myoglobin, which explains why they can stay underwater without breathing for so long. Myoglobin may also play a role in picking up and isolating reactive species of oxygen that are made during cellular respiration.
6. Trypsin
Trypsin is an enzyme that is found mostly in the digestive systems of vertebrates. The main function of trypsin is to facilitate the breakdown of proteins into amino acids. Some amino acids cannot be synthesized in vivo, so the body relies on the activity of trypsin to breakdown protein sources into its constituent amino acids.
Trypsin breaks down proteins by catalyzing the hydrolysis of peptide bonds. Without trypsin, proteins molecules are too large to be absorbed by the large intestine.
5. Tubulin
Tubulin is a family of structural proteins that form the bulk of the microtubules and cell cytoskeleton. The two main kinds of tubulin found in human cells are tubulin-α and tubulin-β. Microtubules are made of cyclical chains of tubulin-α and tubulin-β arranged in a pseudo-helix shape. Tubulin dimers are slightly polar and this polarity is relevant to their functioning in microtubules. The overall polarity of tubulin allows microtubule strands to align themselves in the proper direction, as they are attracted to the polar end of the centrioles.
Aside from providing the main mechanical support of the cell, tubulin also helps with cellular transport. Tubulin molecules in the cytoskeleton serve as “tracks” that other proteins use to move materials through the cell.
4. Globulin
The globulins are a family of proteins that are present in human blood and serve a number of function. Globulin proteins transport materials through blood, initiate blood clotting and regulate blood composition concentrations. Most globulins in blood work in feedback mechanisms to inhibit or stimulate process in the blood.
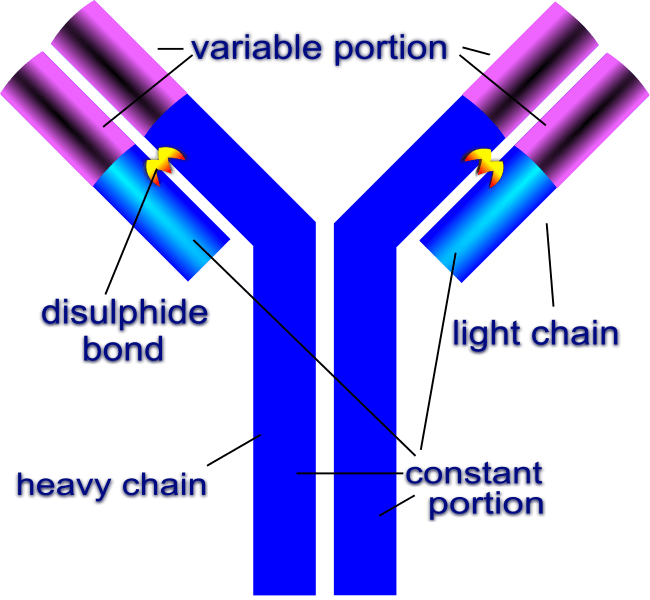
An immunoglobulin molecule. Credit: DigitalShuttermonkey via WikiCommons CC BY-SA 3.0
One particularly important globulin is immunoglobulin. Molecules of immunoglobulin are called “antibodies” and are the main entities that fight against infection. Y-shaped antibodies bond to infectious pathogens by recognizing unique pathogen molecules called antigens. The antibody acts as a “tag” that signals to other cells to attack infectious agents. Antibodies are secreted by B cells in the blood’s plasma.
3. Histone
Histones are proteins that are found in the cell nucleus of eukaryotes. Histones have an abnormally high alkali content. Histone is the main component of chromatin, a nuclear complex of RNA, proteins, and DNA that serve to organize and package nuclear DNA. Histone makes the central column that DNA wraps itself around to form the chromosomes. Unwound, each chromosome contains about 1.8 meters of DNA. With DNA wrapped around the histones, chromosomes are about 90 micrometers in length Aside from physically organizing DNA into chromosomes, histones also play a role in gene expression and regulation.
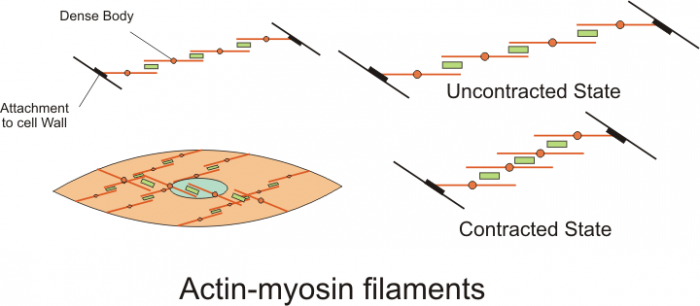
2. Myosin
Myosin is a family of motor proteins that assist with a variety of movement processes in eukaryotes. Most importantly, myosin is the main protein involved in muscle contraction. Myosin proteins bind to actin in eukaryote cells and form long chains. Each myosin molecule can be divided into a head that binds to actin, a middle section that generates the majority of force during muscle contraction, and a tail section that anchors and regulates muscle contraction.
Energy from ATP is used to remove a phosphate group from myosin. The removal causes the shape of the myosin molecule to change, contracting and pulling on actin. Cycles of theses contraction are what result in macroscopic muscle contraction. Muscle contraction cycles can occur as long as there are enough ATP and calcium ions in muscle tissue.
1. Collagen
Collagen is the most abundant protein in the human body, making up around 35% of the body’s whole protein content. Collagen forms the physical structure of most connective tissues in the body, including tendons, ligaments, cartilage, and bone. Collagen is produced by cells called fibroblasts. The exact properties of collagen differ depending on the tissue, though most types of collagen form tough fibrous strands give connective tissue mechanical strength and flexibility. Gelatin found in foods like jello or fruit snacks is derived from collagen.
Collagen has a number of medical applications. Because it is a major component of connective tissue, collagen can be used to stimulate skin growth for wound care and cosmetic surgery. Collagen is also frequently used in bone grafts to help broken bones set and recover.