
Natural killer (NK) cells are fast-acting innate immune cells that have the ability to recognize and kill a variety of cancers. For most healthy individuals not suffering from any rare disorders that render the immune system deficient of NK cells, these super killers are constantly surveilling the body, such that any threat from infection or cancer is quickly eliminated. But how do we still develop cancer despite this diligent immune surveillance?
Cancer cells employ numerous mechanisms to escape NK cell surveillance. For example, cancer cells can escape NK cell immunity by getting rid of cell surface markers that identify them as targets for NK cell elimination. Alternatively, they can gain markers on their cell surface that flash a “STOP” signal to prevent NK cell killing. The process of cancer growth and progression also relies on the recruitment of many factors in the microenvironment that ensure that cancer cells can continue to grow, uninhibited by the immune response.
Cancer immunotherapy utilizes our body’s natural ability to target cancer, by activating and expanding NK cells to enhance their killing ability before infusion into patients. This activation helps NK cells to overcome the “STOP” signals present in the tumor microenvironment. There are two main ways NK cells can be activated to kill cancer cells. The first method uses activating proteins known as cytokines, which promote a general pro-inflammatory response in NK cells. This is the gold-standard in NK cell-based immunotherapy and has been extensively studied and tested in clinical trials since the 1980s. Although it has shown relative success in terms of efficacy, it is associated with a myriad of toxicity problems. An alternative approach to cytokine-based NK cell activation is through exposure to tumor cells. Our research group is interested in the latter, less-studied approach, to better understand how tumor cells activate NK cell immunity.
In this recent study, we aimed to define a tumor-specific signature in human NK cells. NK cells were exposed to a variety of tumor cells or cytokines before characterizing their activation profile. We found that NK cells show a unique pattern of cell surface marker expression after exposure to tumors. We observed a loss in the expression of several NK cell receptors upon interaction with tumor cells (including CD16, NKG2D, and NKp46). Although this pattern is typically associated with NK cell dysfunction in cancer, these “tumor-primed” NK cells show enhanced anti-tumoral effector functions. Previous work by our group and others demonstrated better NK killing of cancer cells after the loss of cell surface markers like CD16. It is possible that this loss is part of the tumor-activation process, contributing to better NK cell surveillance of the tumor microenvironment.
Interestingly, NK cells were faster to secrete pro-inflammatory soluble factors following tumor interactions. Tumor-primed NK cells secreted factors which aid in the activation of an immune response such as interferon-gamma or tumor necrosis factor-alpha within six hours of exposure to tumor cells. In contrast, cytokine-priming of NK cells required longer incubation periods to achieve comparable levels of secretion. This suggests that NK cells may have evolved to respond faster and more potently to contact with a tumor or infected cell, perhaps based on the level of danger it signifies to the host compared to an activating cytokine.
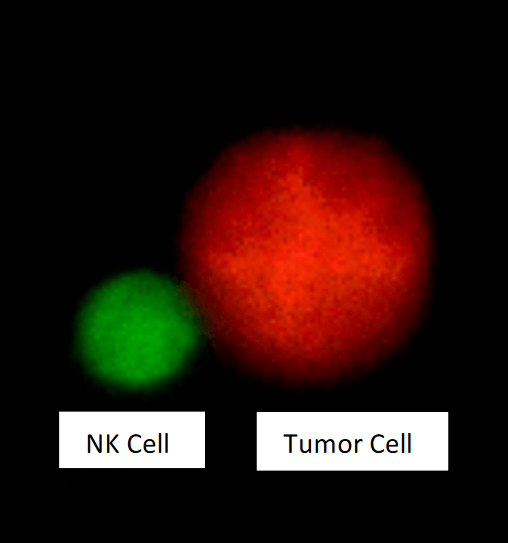
Natural killer (NK) cell in contact with a tumor cell. Freshly isolated human NK cells were pre-labelled with Cell Tracker green and co-incubated for 20 minutes with the leukemic cell line CTV-1 pre-labelled with Cell Tracker Orange, before imaging with a confocal microscope. Figure courtesy May Sabry.
Finally, a unique gene expression pattern was observed in NK cells after exposure to tumor cells. Tumor-primed NK cells showed increased expression of genes specifically linked to NK cell killing of cancer cells (e.g. MAP2K3, STAT5A, TNFAIP3). Distinct molecular pathways triggered in NK cells after interacting with cancer cells may explain the greater cytotoxic activity of tumor-primed NK cells observed in vitro and may lead to greater clinical efficacy compared to cytokine-activation.
Collectively, our results suggest that the interaction between NK cells and tumors generates a signature that highlights the specificity of the NK immune response in cancer. They build on previous findings to challenge the dogma that all activated NK cells look and behave similarly irrespective of the method of stimulation used. In the context of cancer, this NK cell signature might have important implications from prognosis to treatment.









