
If you are ever asked what a polymer of nucleic acid is, know that this is something of a trick question. Nucleic acids are actually polymer themselves. The term polymer simply describes a macromolecule like a nucleic acid or protein. The distinguishing characteristic of polymers is that they are made up of smaller constituent parts, and these sequential parts are referred to as monomers. The monomers are connected together to form polymers. In the case of nucleic acids, the monomers which make up the polymers – the nucleic acids DNA and RNA themselves – are the following: uracil, guanine, cytosine, adenine, thymine.
Nucleic acids are often referred to as “the blueprint of life”, because without these important polymers cells would not be able to grow, replicate, and comprise the variety of life that we see all around us. Let’s take a closer look at how the polymers (nucleic acids) function within the cells, and what role the monomers play in comprising the polymers and carrying out replication.
Defining Nucleic Acids
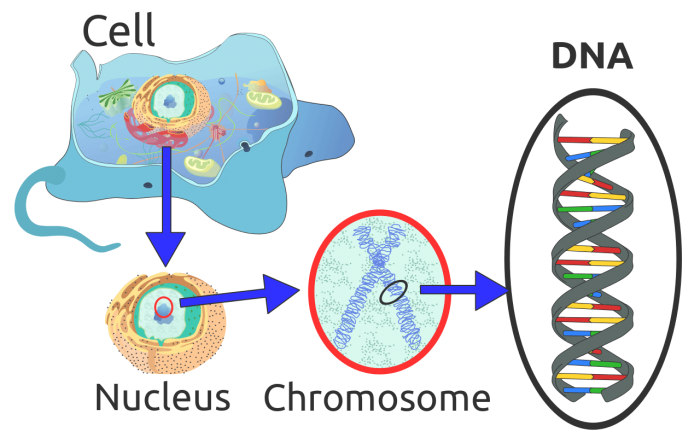
Photo: By Eukaryote_DNA.svg: *Difference_DNA_RNA-EN.svg: *Difference_DNA_RNA-DE.svg: Sponk (talk)translation: Sponk (talk)Chromosome.svg: *derivative work: Tryphon (talk)Chromosome-upright.png: Original version: Magnus Manske, this version with upright chromosome: User:Dietzel65Animal_cell_structure_en.svg: LadyofHats (Mariana Ruiz)derivative work: Radio89derivative work: Radio89 – This file was derived from: Eukaryote DNA.svg:, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=20539140
Nucleic acids are macromolecules, and these macromolecules contain the information necessary for life. Strands of nucleic acid are passed down from parent to child, from one cell to another, and the information within them is used create the proteins necessary for cells, tissues, and organisms as a whole. The two different forms of nucleic acid are ribonucleic acid or RNA and deoxyribonucleic acid or DNA.
All living cells contain both RNA and DNA with the exception of some cells like mature red blood cells. The DNA of the cell is typically found within the cell’s nucleus, though not all cells have the DNA located in the nucleus (hair and nail cells have no nucleus). However, DNA is common to all living things and single-celled bacteria, plants, and animals all have DNA. In contrast, RNA is also used by cells that have DNA, even though not everything that has RNA has DNA. RNA is the primary system of genetic information storage in viruses, though viruses aren’t typically considered to be alive by scientists.
Within the cell, DNA is found not only within the nucleus but in some other organelles such as the mitochondria (a structure which produces energy for the cell). Eukaryotic cells typically have a nucleus with DNA in it, while prokaryotes lack the membrane encased nucleus characteristic of eukaryotes. DNA is contained within long chunks or strands of genes called chromosomes, and every one of these chromosomes has thousands of genes coding for many different proteins. The genes store the genetic information the cell needs to create specific proteins or the “blueprints” for protein synthesizing.
RNA comes into play when it’s time to create proteins. There isn’t just one form of RNA, there are three different forms: ribosomal RNA or rRNA, messenger RNA or mRNA, and transfer RNA or tRNA. The job of the messenger RNA is to create a transcript of the DNA chain, to copy its instructions. This is accomplished by reading the DNA strand and creating a copy of every DNA sequence. Afterward, the transfer RNA will take the code produced by the mRNA and transfer of amino acids to the ribosomes so that proteins may be synthesized. Finally, the rRNA helps produce ribosomes themselves.
The Nucleotides And Nitrogenous Bases
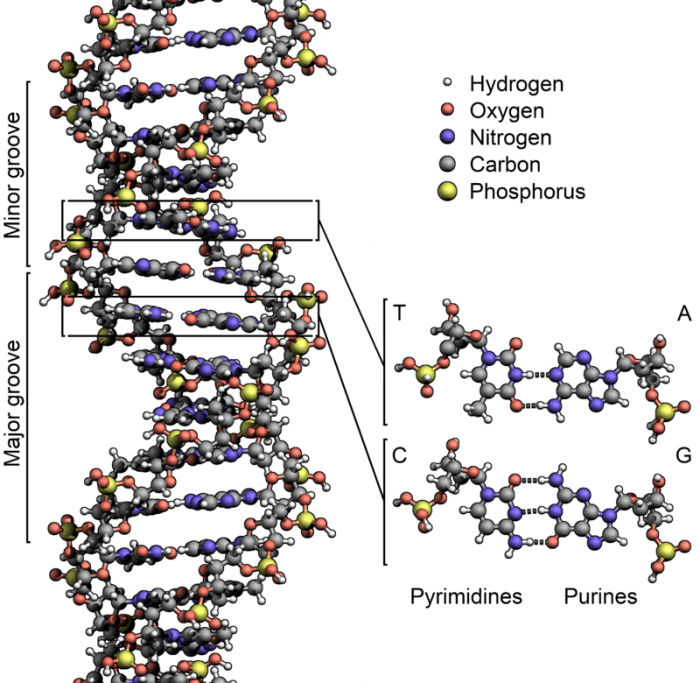
Structure of DNA. Photo: By Zephyris – Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=15027555
As previously discussed, RNA and DNA are polymers, meaning that they are made out of simpler components called monomers. Nucleotides are the term for the DNA and RNA themselves, the complex polymers. Nucleotides can be combined together to create polynucleotides. Every nucleotide possesses the same general structure, a nitrogenous base ( a ring structure made out of nitrogen), a phosphate group, and a five-carbon sugar. The carbon and phosphate groups branch off of the central sugar molecule.
The monomers that comprise DNA and RNA are referred to as nitrogenous bases, the carbon-based molecules located within the ring structure in the nucleotides. DNA has four nitrogenous bases that comprise it: guanine, cytosine, thymine, and adenine. These are abbreviated as just G, C, T, and A. Because guanine and adenine have two carbon-nitrogen rings fused together, they are called purines. Meanwhile, a single nitrogen-carbon ring forms cytosine and thymine, and they are referred to as pyrimidines.
RNA has a different structure than DNA, with one of the major differences being that it lacks the nucleotide thymine. Instead, RNA has a different pyrimidine that takes thymine’s place, a nucleotide called uracil. RNA still possess the acids guanine, adenine, and cytosine, however.
Other differences between DNA and RNA include the fact that the two molecules have not only different bases but different sugars. The sugar that makes up DNA is called deoxyribose while the sugar that constitutes RNA is just a ribose. While the two sugars are quite similar to one another, there is an important difference. The second carbon in deoxyribose has hydrogen, while the second carbon in ribose has a hydroxyl group.
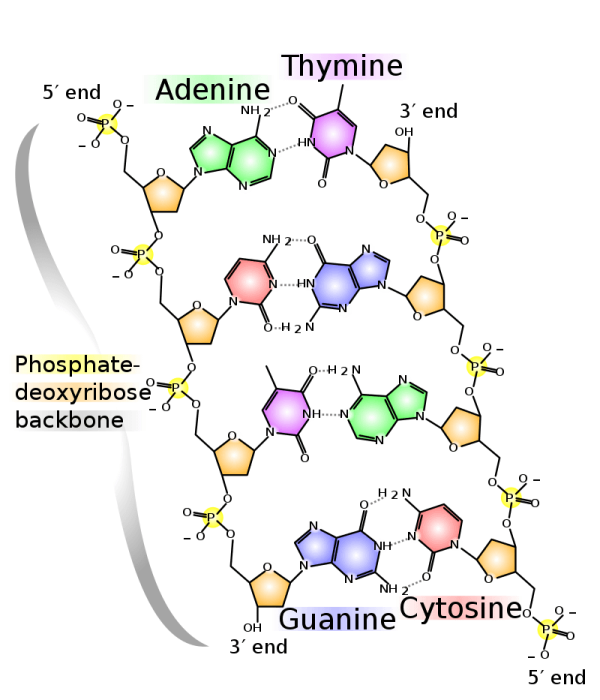
Photo: By Madprime (talk · contribs) – Own workiThe source code of this SVG is valid.This vector image was created with Inkscape., CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=1848174
The center of a nucleotide is comprised of the sugars while the phosphates are links to the 5’ carbon position and the nucleotides joined to 1’ carbon position. The phosphate groups in nucleotides can either be multiple phosphate groups or a single phosphate group. Up to three phosphate groups can be joined to nucleotides at the 5′ carbon sugar point.
You should be aware that some resources utilize the term nucleotide to describe only the bases that are linked with a single phosphate group, so there is some disagreement over what exactly counts as a nucleotide. It’s also important to remember that the creation of a polynucleotide, when DNA and RNA chains join together, results of the loss of two phosphate groups. This means that if there is a chain of DNA or RNA molecules, the molecules will only have a single phosphate group.
Molecules That Make Up Nucleic Acids
What are the individual molecules that compose monomers and by extension, compose polymers? Five different molecules are combined in different ways to create nucleic acids: oxygen, hydrogen, carbon, nitrogen, and phosphorus. Each one of these molecules has its own role to play, creating different parts of the RNA/DNA molecule.
Carbon molecules are critical for the creation of nucleotides, as they not only create the nucleic acid backbone of the molecule by comprising the sugar in the backbone, they also comprise parts of the nitrogenous bases. Both pyrimidines and purines, the two forms of amino acids in nucleotides, are created by nitrogen molecules. There are hydrogen bonds found between amino acid groups, which enables the base pairs to stay linked together in the strands of RNA and DNA.
Hydrogen molecules link the carbon and oxygen atoms between the sugars of the nucleic acids and the nitrogenous bases. These nitrogen-hydrogen bonds are polar in nature, and they help hydrogen bonds link entire strains of nucleic acid together. These hydrogen bonds are how the double helix formation that DNA is known for is created since the two strands of DNA are linked together through the base pairs’ hydrogen bonds. The phosphates, nitrogenous bases, and sugar also have oxygen atoms interspersed within them. The presence of these oxygen atoms affects DNA and RNA in slightly different ways, so DNA and RNA have different sugar structures. Even though ribonucleic acid has four hydroxyl groups, this isn’t true of DNA which has a pure hydrogen stand-in for one of the hydroxyl groups. This is how the “deoxy” in the term and deoxyribonucleic acid is applied.
Finally, phosphorus molecules join together to construct the phosphate groups found within both RNA and DNA. Both phosphorus and oxygen atoms are necessary to create these groups. Phosphorus groups enable different nucleotides to join together to create polymers.
Difference Between Nucleotides And Nucleosides
Nucleotides sound similar to nucleosides, but the two what should not be confused. What is the difference between nucleosides and nucleotides? The short answer is that nucleosides are very similar to nucleotides except they lack a phosphate group. Nucleotides are created from and nucleosides, undergoing a process known as phosphorylation to become nucleotides. The phosphorylation process has nucleosides and phosphorus join together to make a nitrogenous base.
The cells of the body can create nucleosides through synthesis, but eating food can also provide the body with nucleotides. The nucleotides acquired by eating food Are degraded by an enzyme called nucleotidase, and broken down into nucleosides and phosphates.
Codons And Protein Synthesis
RNA strands are divided into three letter or molecule long sequences, and these sequences specify features necessary to create the correct proteins. The term for these three letter codes is codons, and they can be made out of any combination of the three nucleotide bases of RNA. The codons are read by ribosomes, and the ribosome will interpret the code on sequences To create the correct proteins. Protein synthesis is started by the presence of a start codon, the codon AUG or methionine. When this codon is red, the ribosome brings transfer RNA into it and begins synthesizing proteins by reading of the tRNA sequences and applying the necessary amino acids and anti-codons, the complementary sequences to the codons the mRNA has delivered.
Within the strands of RNA, there are 64 total codons. As mentioned, the codon AUG is the start codon. There are also three different stop codons, which leaves 61 other codons that can be combined to create a variety of different proteins. Humans not only utilize 20 of the different codons to create amino acids, and multiple codons can code for the same protein, such as the protein leucine being coded for by UUG and UUA.









