
Scientists typically refer to the rise in global average temperature and the associated effects on Earth’s climate with the term “climate change.” Lately, it is becoming clear to everyone that the cause of climate change is the ever-growing energy demand to power humans’ modern lives. As a matter of fact, conventional power generation systems burn fossil fuels to produce energy, with the consequent release of a large amount of greenhouse gases in the atmosphere. This extra amount of heat-trapping gases is the acknowledged cause of the rise in temperatures.
In the last decades, many scientists focused their research on power generation systems potential alternatives to those running on fossil fuels to limit the worsening of climate change. In the automotive sector, Proton Exchange Membrane Fuel Cells (PEMFCs) represent one of the most promising alternatives to conventional internal combustion engines thanks to their high specific power and efficiency, quick refueling time, fast warmup and shut down, as well as low noise and zero pollutant emissions.
The performance and consumption specifications of the main fuel cell vehicles available on the market are reported in Table 1 [1,2,3]. The cost per mile of these cars is calculated with respect to the current market price of hydrogen, which is around 15 $/kg [4]. On the contrary, the cost per mile of the average internal combustion engine vehicle is much smaller than the previous ones: it is around 0.07 $/mile if 0.08 liter fuel consumption is assumed per mile and a cost of 0.9 $ per liter of fuel. Anyway, this situation can change significantly in the near future.
In fact, thanks to the improvement of hydrogen refueling infrastructures and of production systems, hydrogen cost per kilogram is expected to go down to 8 $/kg in the next decade. In this scenario, fuel cell vehicles would become cheaper to run than the internal combustion engine ones, especially in those areas where the oil price is higher due to geographical location or economical duties (in Europe, for example, oil price is almost doubled compared to the United States [5]).
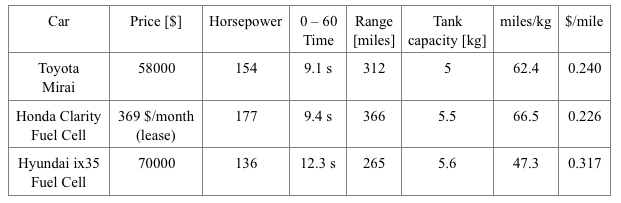
Table 1 – Performance and consumption specification of fuel cell vehicles. Credit: Alessandro Ferrara
Despite their running costs, several drawbacks are hindering PEMFC commercialization on a large scale, such as their limited durability and their high manufacturing and maintenance costs. For this reason, scientific and industrial efforts are currently oriented toward costs reduction and durability increase. In particular, it is possible to increase the durability and reliability of the system thanks to suitable control strategies to avoid (or at least limit) the occurrence of detrimental operating conditions, which negatively affect system performance and lifetime.
A PEMFC is an electrochemical device that directly converts hydrogen chemical energy into electricity, heat, and water. Since there is no combustion within the cell, the system is not restricted by the limitation imposed by Carnot’s principle and, hence, it can achieve higher efficiency with respect to a conventional power generation system. The cell is made by two platinum-based electrodes (anode and cathode) detached by a selective polymeric electrolyte that allows only proton and water flows. The inlet hydrogen reacts on the surface of the platinum catalyst at the anode and, as consequence, electrons are separated from protons: electrons are collected in an external circuit, generating the electrical output of the cell, while protons pass through the electrolyte. Hence, through distinct paths, both protons and electrons reach the cathode, where they are combined (on the surface of the platinum catalyst) with the inlet oxygen to produce water (zero emissions).
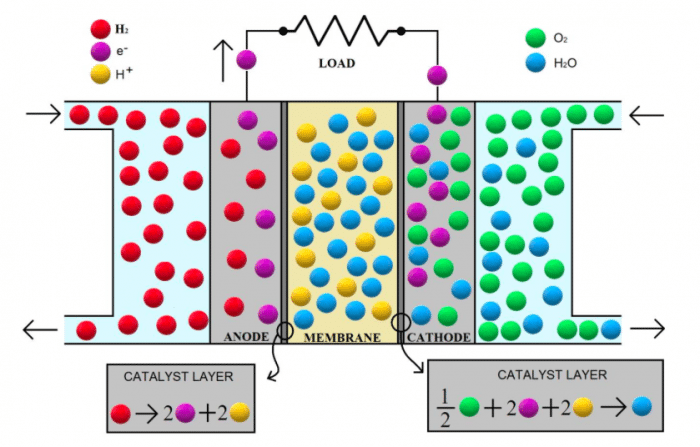
Figure 1 – Proton Exchange Membrane Fuel Cell working principle schematization. Credit: Alessandro Ferrara
One of the most critical issues related to PEMFC operation consists in its water content management. A correct electrolyte hydration promotes the proton transport from anode to cathode, resulting in fewer cell losses. Therefore, if the electrolyte water content is reduced, proton transport resistance increases, with a consequent voltage drop. This phenomenon is known in the literature as drying.
On the other hand, if the water content increases too much, although the electrolyte conductivity is enhanced, the presence of a great amount of liquid water hinders the reactants (i.e. oxygen and/or hydrogen) from reaching the reaction sites. This leads to a growth in the diffusion losses and to a consequent voltage reduction as well. This phenomenon is usually called flooding. Accordingly, it is clear the importance of real-time diagnostic and control strategies (based for example on mathematical models representative of fuel cell physical phenomena) that keep electrolyte water content within an optimal range, to balance the occurrence of the aforementioned detrimental phenomena.
The use of models for real-time simulations is only possible if the required computational effort is suitable for on-board devices. For this reason, the authors of this paper stressed the need for fast-simulation and proposed a simplified mathematical model of the overall water transport through the electrolyte to estimate the electrolyte water content of a PEMFC as a function of electrolyte physical properties and cell operating conditions (i.e., current, temperature, reactants humidity, etc.).
The model design is chosen as a trade-off between computational speed and accuracy, with great attention to the correct representation of the involved water transport mechanisms (i.e., electro-osmosis, diffusion, convection, and swelling). In particular, a fast approximated analytical solution of the mass conservation equation is preferred to the exact solution that is, instead, time-consuming. The analytical solution accuracy is assessed with respect to the exact solution achieved through a numerical approach (which requires greater computational effort and time). The obtained results show that, for the operating condition in which the cell usually works, the maximum and mean approximation errors are limited to 0.8% and 0.26%, respectively.
According to its design, the model guarantees high simulation accuracy while offering the possibility to be easily embodied into more complex frameworks (cell or stack models), for fuel cell simulation, on-board/real-time monitoring, diagnostic, control, and prognostic algorithms. In such a way, the model may support the development and application of suitable strategies to detect in reasonable time drying and flooding and apply proper countermeasures to reduce the detrimental effects of these phenomena on cell performance and durability.
For further details, the readers are directed to the related work titled, Analytical calculation of electrolyte water content of a Proton Exchange Membrane Fuel Cell for on-board modelling applications, with authors Alessandro Ferrara, Pierpaolo Polverino and Cesare Pianese (Dept. of Industrial Engineering, University of Salerno, ITALY) published in the Journal of Power Sources, Volume 390, Year 2018, Pages 197-207 (0378-7753/ © 2018 Elsevier B.V. All rights reserved).
References:








