Centrosome is an organelle responsible for the organization of microtubule arrays, typically implicated in the organization of cell division, cell motility, and cell shape. The core of the centrosome is made by a pair of centrioles, barrel-shape structures embedded in a matrix of proteins termed the pericentriolar material. As centrosome function is tightly linked to cell cycle, it comes as no surprise that centrosome biogenesis is under the watchful eye of the cell cycle machinery.
Centrosome duplicates once per cell cycle, with proteins PLK4 and STIL playing the pivotal role in the regulation of a key step in centrosome cycle – the duplication of centrioles. As the cell divides, each daughter cell inherits precisely one centrosome. Even though centrosome and centrioles have already been studied for over a century, it is only the recent years when centrosome-focused research has boomed and flourished, as many previously unpredicted links to human diseases, embryogenesis, and development have been revealed.
Human pluripotent stem cells, comprising both embryonic stem cells and induced pluripotent stem cells, possess two remarkable features: pluripotency — the ability to differentiate into a wide variety of cell types of the adult organism — and self-renewal — the ability to indefinitely divide and produce cells with unchanged potential. These properties have attracted a lot of attention during last years and made stem cells a subject of increasing scientific interest because of their potential utility in numerous biomedical applications, such as excellent in-the-dish model to study events important for human embryogenesis. Curiously, despite numerous studies with these cells, the importance and function of centrosome in this cell type has remained elusive.
In the study Inactivation of PLK4-STIL Module Prevents Self-Renewal and Triggers p53-Dependent Differentiation in Human Pluripotent Stem Cells, Renzova and colleagues blocked the function of PLK4 or STIL with the idea to put a brake on the centrosome duplication pathway and hence to prevent centrosome to duplicate in stem cells. The rationale behind this experiment was that with centrosome duplication being blocked, stem cells may attempt to divide even when having only one centrosome. In case they succeed, the centrosome will be inherited only by one daughter stem cell; the second daughter will be without any centrosome. In a situation where even cells with one or no centrosome manage to divide this should, with time, ultimately lead to the increased emergence of stem cells without any centrosomes. See also the scheme of the experimental paradigm (Figure).
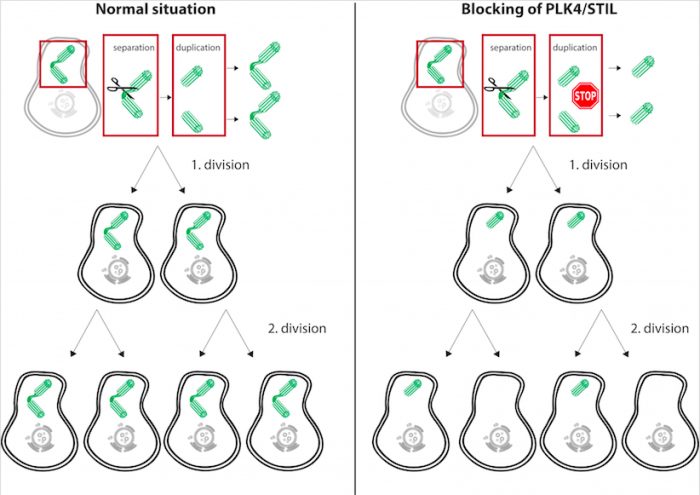
Credit: Ondřej Bernatík
The study revealed that applying this strategy led to a complete depletion of centrosome in over 85% of stem cells after 3 days of blocking the duplication pathway. Most of the subsequent analyses were then conducted at this time point, and revealed several interesting outcomes. Firstly, analysis of cell divisions confirmed that stem cells are able to arrange mitotic spindle and to divide even without any centrosome, with no major impact on survival of these cells, which was not anticipated. In fact, these findings raised an alerting question if centrosome might perhaps be a completely obsolete organelle for human stem cells. However, subsequent in depth analyses dismissed this possibility, showing that stem cells do pay a price for losing their centrosome. Namely, the mitosis in the absence of centrosomes took much longer, and was prone to errors, which led to a prominent increase of chromosome abnormalities (aneuploidy). Importantly, self-renewal potential of these cells was strongly compromised, the cells stopped proliferating 4-5 days after the centrosome duplication block was applied.
The team subsequently focused on the examination of effects of centrosome depletion on stem cell properties to find that the centrosome loss led to downregulation of regulators of pluripotency OCT-4 and NANOG and a concomitant increase in the expression of proteins which marked the initiation of differentiation program (namely p53, PAX-6, brachyury etc.). Given that p53 has been shown to mediate consequences of centrosome loss in mice and human somatic cell lines, the team examined its role in the context of centrosome loss in human stem cells. The experiments revealed that p53 was activated due to the prolonged mitosis following centrosome loss, but the molecular identity of putative stress signal leading to its upregulation was not deciphered yet.
Unexpected results came out of rescue experiments, which were done to assess how much is p53 responsible for the defects observed in stem cells after losing centrosome. The work revealed that p53 mediated effects of centrosome depletion in stem cells only partially – removing p53 prevented induction of differentiation markers and led to partial restoration of self-renewal capacity, but the effect of centrosome loss on downregulation of pluripotency regulators such as OCT-4 was still present. This suggested that additional effector(s) contribute to the centrosome loss phenotype. Further experiments subsequently pointed on altered protein turnover as the one been guilty here – cells depleted of centrosome degraded some of the pluripotency proteins faster than controls.
To conclude, Renzova et al. linked centrosome homeostasis to effective self-renewal of stem cells. This study identified both p53-dependent and -independent consequences of centrosome loss in stem cells, connected them to alterations in mitotic timing and protein metabolism and provided insight on how centrosome abnormalities may contribute to a pathology of centrosome-related diseases.
These findings are described in the article Inactivation of PLK4-STIL Module Prevents Self-Renewal and Triggers p53-Dependent Differentiation in Human Pluripotent Stem Cells in the journal Stem Cell Reports. This work was conducted by Tereza Renzová, Dáša Bohačiaková, Milan Ešner, Veronika Pospíšilová, Tomáš Bárta, Aleš Hampl, and Lukáš Čajánek at Masaryk University, Faculty of Medicine. We are grateful to Ondřej Bernatík for help with preparation of the figure.









