Malaria parasites can tell good times from bad times and plan their offspring accordingly. The reproductive strategy these disease-causing parasites use is more sophisticated than previously thought and similar to that of more complex organisms like mammals, birds, insects, and plants.
Malaria parasites (Plasmodium) are small blood parasites that are transmitted between hosts by mosquitoes. The parasites replicate (divide) in the red blood cells of their host, which allows the parasites to survive in the host but also causes disease symptoms. To infect new hosts, the parasites have to make specialized sexual stages that infect the mosquito vector, subsequently leading to the infection of new hosts. It is important for the parasites to balance how much they invest in replicating blood stages for survival in the current host versus sexual stages for transmission to a new host. At the same time, this balancing act influences clinical disease outcomes and how well the disease spreads.
In recent years, scientists have learned more about the biological processes inside the malaria parasite that lead to the formation of sexual transmission stages. However, it is not clear why these processes occur in only a small proportion of the parasites in a host, and this proportion is variable between host and during infections. In other words, we do not yet understand how parasites “decide” how much to invest in sexual transmission stages. A theory developed for multicellular organisms predicts that the circumstances an organism lives in should determine how much they invest in reproduction. We applied this theory to malaria parasites.
We used drug treatment as a method to manipulate parasite survival in the host and tested how parasites respond with respect to their investment into survival versus reproduction. We first used a mathematic model to predict how parasites should respond to changes in their survival rate, and we check that a flexible strategy for investment in reproduction indeed benefits the parasites (as opposed to always sticking with one and the same strategy). We then tested our predictions from the model using rodent malaria parasites (Plasmodium chabaudi) in mice. Infections were treated with one of a range of drug doses and we calculated the investment in sexual transmission stages.
We discovered that the parasites change how much they invest in survival versus reproduction according to the conditions they experience during infection. More specifically, when parasites experience good conditions and can grow well within the host, they can afford to reproduce and invest in spreading to new hosts. When times are bad and parasite survival is threatened, parasites delay reproduction. This way they can divert efforts into replicating themselves, prioritizing survival in the host – a strategy we call “reproductive restraint.” But if conditions are catastrophically bad, and the parasites have very little chance of surviving in the host, they invest as much as possible in reproduction in a “last-ditch” effort to spread to new hosts. This strategy is what we call “terminal investment.”
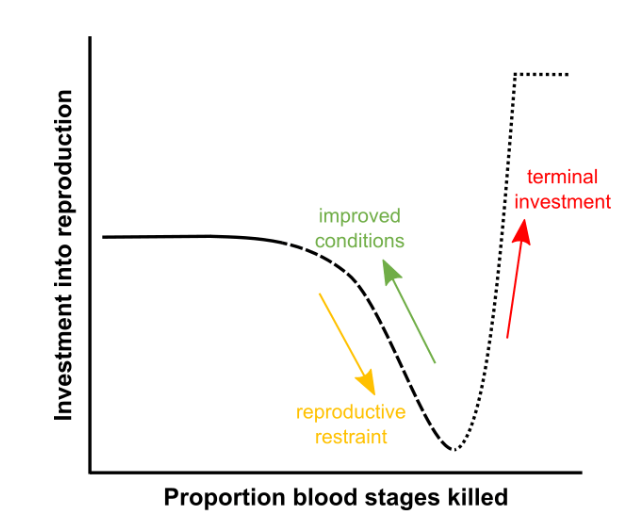
Figure 2. Changes in reproductive strategy in response to a loss in survival. Malaria parasites that experience good conditions in the host and are growing well can afford to invest in sexual transmission stages (solid line). If their survival deteriorated they adopt reproductive restraint (dashed line), and they switch to making a terminal investment (dotted line) if their survival in the host becomes unlikely. Image modified and republished with permission from PLOS Pathogens from https://doi.org/10.1371/journal.ppat.1007371.
It has been known for a long time that malaria parasites in stressful conditions can prioritize reproduction over survival in the host (terminal investment). This was assumed to be their standard response to stress. However, data were inconsistent, and more recently we showed that malaria parasites can also prioritize survival over reproduction (reproductive restraint) when conditions are only marginally stressful. Our current work shows that these two opposite strategies are actually part of one continuum, a sophisticated reproductive strategy similar to that of mammals, birds, insects, and plants.
We now know that malaria parasites are flexible in their investment into survival versus reproduction and that how much they invest in sexual transmission stages depends on conditions in the host. Parasites make their reproductive decisions based primarily on parasite survival, but then modulate their response depending on whether red blood cell resources are improving (i.e. recovering from anemia) or deteriorating (i.e. anemia becoming worse). Being able to predict how the parasites respond to conditions that kill them, including drug treatment, is important because reproductive restraint (prioritize survival over reproduction) can make infections harder to clear and terminal investment (prioritize reproduction over survival) can help to clear infections whilst increasing the risk of infecting mosquitoes and subsequent hosts.
Understanding how parasites react when we try to kill them may open up new ways to harness this parasite response in a direction that is clinically favorable. For example, developing treatments that prompt the parasites to boost reproduction while also blocking the spread to new hosts (e.g. with bednets) could help combat the disease.
These findings are described in the article entitled Adaptive plasticity in the gametocyte conversion rate of malaria parasites, recently published in the journal PLOS Pathogens.
Note
This work was supported by NERC, BBSRC, the Royal Society, the FNR of Luxembourg, the Wellcome Trust, the Human Frontiers Science Program, and the Natural Sciences and Engineering Research Council of Canada.









