Cells have been the building block of all organisms on Earth ever since they presumably emerged more than 3.5 billion years ago. Scientists have invented several methods to discover and study the structure and function of new cell types. Among them, microscopy has particularly been an essential tool for studying cells since Robert Hooke, who is credited for coining the term cell, discovered them in 1665.
Approximately three centuries later, flow cytometry emerged, enabling rapid analysis of individual cells at a high throughput of tens of thousands of cells per second by measuring light emissions from flowing cells; the method has dramatically evolved since Mark Fulwyler and Leonard Herzenberg introduced cell sorting. Another essential method includes molecular analysis, including genomic and transcriptomic analysis using next-generation sequencing (NGS).
Despite the effort put into studying cells, scientists still fail to unfold some of the critical biological phenomena, demanding further improvement of research methods. Exploring such unknown phenomena can lead to addressing issues related to our daily life, such as eliminating cancer cells, creating functional organs for transplantation, and substituting algae-produced oil for fossil fuels. To advance these studies, scientists have been combining different modalities: cell sorting bridges flow cytometry and molecular analysis by purifying a population of cells identified by flow cytometry for transcriptomic analysis, while imaging flow cytometry combines flow cytometry with microscopy. Ultimately, scientists dream to integrate all of the aforementioned methods, microscopy, cytometry, and molecular analysis, to gain thorough information from tens of thousands of individual cells.
In the paper recently published in Cell entitled “Intelligent Image-Activated Cell Sorting,” Professor Keisuke Goda and his colleagues at the University of Tokyo introduce a new method that fulfills such a dream by fusing microscopy and cell sorting (Figure 1).
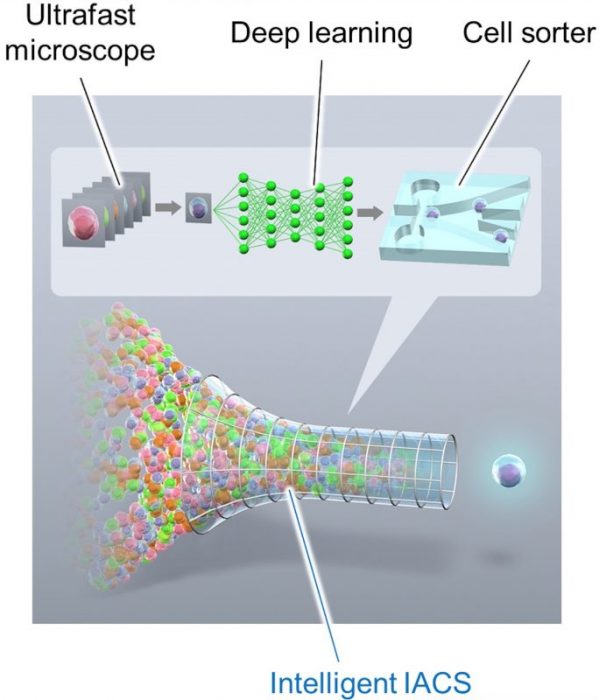
Figure 1. Schematic of the intelligent image-activated cell sorter (intelligent IACS). Image published with permission from Cell from https://doi.org/10.1016/j.cell.2018.08.028.
The intelligent image-activated cell sorter (intelligent IACS) rapidly takes images of cells flowing in a microchannel using a custom-made ultrafast imaging technique. Then, the intelligent IACS analyzes the captured images while the cells are still flowing in the microchannel. Finally, it isolates cells using a micromechanical sorting device. The sort decision is based on multiple image analysis algorithms including deep learning.
The sensing, sorting, and calculating elements communicate with each other via a 10-Gbps all IP network that enables a fast and highly flexible platform for real-time image recognition and decision making for sorting. The sorted cells are ready for downstream analysis including molecular analysis, single cell cloning, and functional assays.
To demonstrate the practicality of the intelligent IACS, the researchers sorted microalgae based on their intracellular protein localization, which is related to the carbon-concentrating mechanism (CCM). The CCM is considered as the turbo for photosynthesis because it plays an important role in boosting the carbon fixation process in algae. Therefore, investigation of the CCM would allow us to engineer the photosynthesis of plants to improve its efficiency. Unfortunately, conventional cell sorters are insufficient because they fail to measure the spatial distribution of proteins, which is vital for the CCM. Additionally, conventional microscopy requires impractical time and effort to study a large number of cells.
To overcome these issues and to accelerate the study, the researchers used the intelligent IACS to sort out mutants with a distributed CCM pattern from a population of randomly mutated algae. Specifically, they screened ~200,000 cells in an hour and isolated ~2,000 cells that might be useful for finding critical genes that regulate the CCM (Figure 2A).
To further demonstrate the versatility of intelligent IACS, the researchers sorted aggregated platelets from human blood samples. The platelet is an essential blood component to stop bleeding, but it can undesirably activate to form blood clots that cause thrombosis. Hence, studying the platelet activation would lead us to diagnosis, drug discovery, and therapeutic monitoring of heart attack and stroke due to thrombosis. However, conventional methods, including flow cytometry, fail to quantify the platelet activation in the body because platelet easily activate while staining the sample, which is a necessary step during sample preparation. Here, the platelet aggregation is a known activation biomarker of platelets measurable with the microscopy included in the intelligent IACS without staining. Thus, the researchers demonstrated in the paper that the intelligent IACS is capable of finding and isolating aggregated platelets, even small aggregates of as few as two cells (Figure 2B).
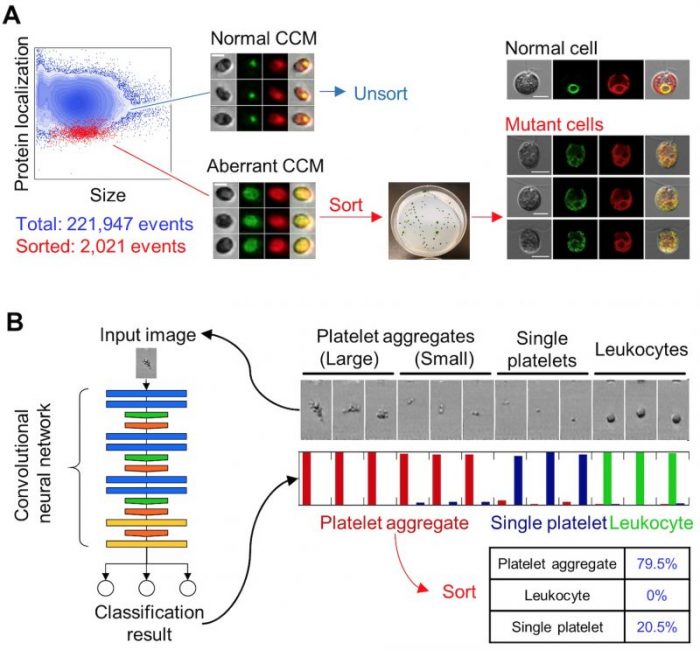
Figure 2. Cell sorting experiments using the intelligent IACS.
A. Enrichment of microalgal mutants. An essential protein in the carbon-concentrating mechanism (CCM) is labeled with a green fluorochrome, which works as an indicator for identifying mutants with aberrant CCM. The identified mutants were sorted, cloned, and verified using confocal microscopy. B. Isolation of platelet aggregates. An image classifier based on deep-learning is employed to recognize platelet aggregates. Sorting of the platelet aggregates followed by microscopic enumeration showed a high sorting purity of 79.5%. Image published with permission from Cell from https://doi.org/10.1016/j.cell.2018.08.028.
The intelligent IACS is expected to open up a new arena in cell biology. Professor Keisuke Goda and his colleagues are offering researchers worldwide an open innovation platform, which provides access to the intelligent IACS and encourages others to test their ideas and samples. They also launched a startup CYBO, Inc. to develop, manufacture, and deliver commercialized products based on the intelligent IACS.
These findings are described in the article entitled Intelligent Image-Activated Cell Sorting, recently published in the journal Cell. This work was conducted by Keisuke Goda and colleagues from the University of Tokyo.
Reference:
- Nitta et al., “Intelligent Image-Activated Cell Sorting,” Cell 175, 266-276 (2018) https://www.cell.com/cell/fulltext/S0092-8674(18)31044-4.








