
Nowadays, conjugated organic polymers have various applications in the field of low-cost and flexible optoelectronic devices [1-5] viz. organic field effect transistors (OFETs), organic light-emitting diodes (OLEDs), photovoltaic solar cells, p-n unction diodes, etc. [6-8]. It is mainly due to their attractive optical, electronic, and electrochemical properties [9,10]. They also have great advantages over the conventional inorganic optoelectronic devices such as low-cost synthesis, long-term stability, low weight, and easy manufacturability of thin film devices, etc. [11-13].
In the last decades, significant improvement of optoelectronic properties in the field of conjugated organic polymers has taken place due to the alternating sequence of donors (electron-rich) and acceptors (electron-deficient) (D-A unit) [14,15]. This alternating D-A approach in organic polymers leads to a significant lowering of the fundamental gap (ΔH-L) of the conjugated organic polymers [16]. The fundamental gap can be defined as the energy difference between the HOMO and the LUMO energy level [17,18].
In general, the conjugated organic polymers undergo change transfer phenomena via hopping type mechanism. However, most of the organic polymers are mainly of p-type. Hence, in this case, the charge transfer process is predominated by the hole-transfer process. The hole-transport in the organic semiconductor materials between adjacent, spatially-separated molecules can be described through self-exchange reaction, expressed [19,20] in eq. 1.
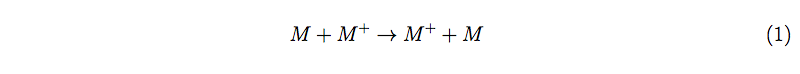
Here, M represents the neutral species which undergoes charge transfer, and M+ is the charged species which contains the hole. The hole transfer rate is primarily dependent on the reorganization energy (λ) due to geometric relaxation accompanying charge transfer and the electronic coupling matrix element (V) between the two species [19]. V is related to the band width in conventional sold state descriptions [19]. According to the tight-binding approximation, the band width is twice the orbital energy splitting observed in a dimeric pair, or four times the charge resonance integral (β) [19]. In literature, it has been reported that orbital energies and band widths can be calculated using semi-empirical methods and, hence, the consequences for charge transfer rates can be understood [19,21].
The hole/electron transfer rate for higher temperature is primarily dependent on the reorganization energy (λ) due to the geometric relaxation [19]. The importance of λ in charge transfer processes has been widely studied in the context of Marcus theory. The hole transfer rate for charge transfer processes can be calculated with the help of the Marcus-Hush model [22-25]:
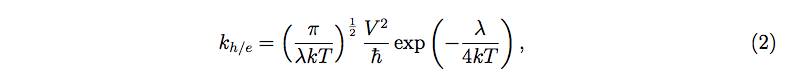
where T is the absolute temperature, V is the electronic coupling matrix element between two species, k is the Boltzmann constant, and ħ is the reduced Planck’s constant. λ needs to be small for efficient charge transport. The electronic coupling matrix element (V) between two species, often called the transfer integral, depends on the orbital overlap between the species and needs to be larger for efficient mobility [26,27].
For a better understanding of the conductivity in conjugated organic materials, it is important to identify the structural factors essential for the charge transfer rates. It has already been reported that the solid-state hole/electron mobility in aryl amine is related to the internal reorganization energy (λ) [29-32]. The internal reorganization energy (λ) of the isolated active oligomers or polymer for hole/electron-transfer in eq. 2 can be expressed as follows:
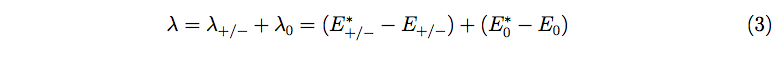
where E*+/- and E*0 represent the energy of the charged species at the optimized geometry of the neutral species and the energy of the neutral species at the optimized geometry of the charged species, respectively. E+/- and E0 stand for the energy of the charged state in the optimized geometry of the charged species and the energy of the neutral state in the optimized geometry of the neutral species, respectively.
In a similar manner, the coupling matrix element is calculated by employing the following expression [33-35]:
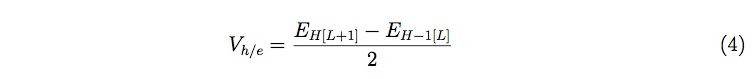
Here, EH and EH-1 represent the energies of HOMO and HOMO-1 orbitals of the molecular systems, respectively.
We have designed a novel donor-acceptor backbone, which contains a triazole moiety linked with a bridged dithiophene moiety. Here, the triazole (Tr) moiety acts as an electron-deficient unit (acceptor), and the bridged dithiophene (DT) moiety acts as an electron-rich (donor) unit [7,36]. The three nitrogen atoms in the triazole ring impart the electron-deficient character to the ring. The DT moiety acts as an electron-rich donor due to the presence of electron-rich sulfur atoms. Sequencing the donor-acceptor moieties in an alternating manner, a significant lowering of the ΔH-L values will be achieved. The conjugated organic polymers possessing lower ΔH-L values generally lie in the range of 1 to 4 eV [37].
The donor-acceptor approach facilitates the reduction of the fundamental gap due to the generation of a molecular packing having large π-orbital overlap. As a result, efficient charge injection occurs across the polymer backbone, making it suitable for the fabrication of optoelectronic devices [15]. Moreover, the λ and V for all the oligomers can be calculated with the help of eq. 3 and eq. 4, respectively, to explain the charge transfer of each oligomer.
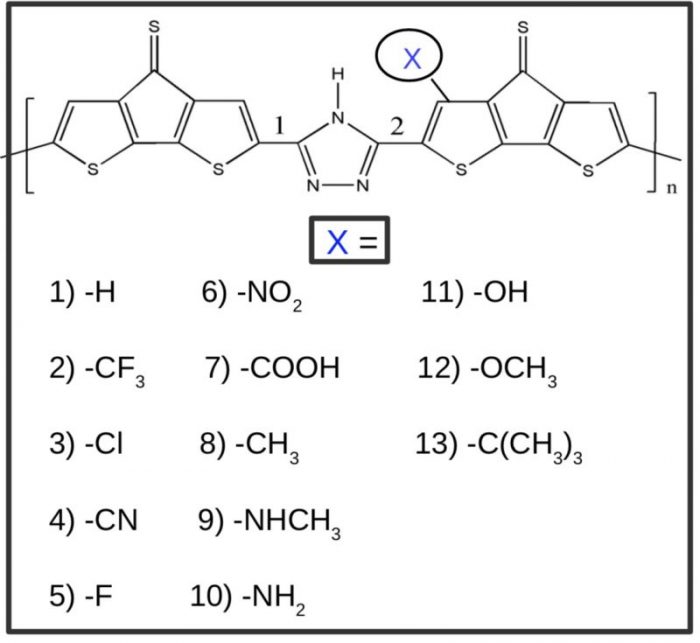
The work is based on thirteen conjugated oligomers. The sketch of the basic unit of the bridged dithiophene-triazole-bridged dithiophene (DT-Tr-DT) based copolymers, along with the thirteen different substituents, are presented in Fig. 1. In the figure, compound 1 represents the basic unit (DT-Tr-DT). From compounds 2 to 7, the DT-Tr-DT unit is substituted with the electron-withdrawing groups viz. (X = CF3, Cl, CN, F, NO2, COOH). Similarly, from compounds 8 to 13, the basic unit is substituted with the electron-donating groups viz. (X = Mr, NHMe, NH2, OH, OMe, tBU).
Credit: Dhruba Jyoti Kalita
In this work, a series of cyclopentadithiophene-4-thione and triazole-based organic oligomers with different substituents are designed by using the donor-acceptor (D-A) approach. We have performed theoretical calculations with the DFT and the TDDFT level of theory incorporating B3LYP, CAM-B3LYP/6-31G(d) functional and basis set. We have also calculated reorganization energies and hole transfer rates.
Our study unveils that the oligomers under consideration exhibit excellent hole mobility rates as well as low ΔH-L values in the range of 1.20 to 1.50 eV. A trimer of compound 11 shows the lowest ΔH-L values (1.28 eV) among the trimers. However, oligomers with especially small electron-donating groups (compounds 8, 10, and 11) will act as potential candidates for optoelectronics. In short, we infer that these organic oligomers will act as interesting materials for fabrication of OLEDs, OFETs, photovoltaic cells, and solar cells [38].
These findings are described in the article entitled, Charge injection and hopping transport in bridged-dithiophene-triazole-bridged-dithiophene (DT-Tr-DT) conducting oligomers: A DFT approach, recently published in the journal Computational and Theoretical Chemistry. This work was conducted by Rakesh Dutta and Dhruba Jyoti Kalita (corresponding author) from Gauhati University.
References:
- W. Zhong, J. Liang, S. Hu, X.-F. Jiang, L. Ying, F. Huang, W. Yang, and Y. Cao, Macro- molecules 49, 5806 (2016).
- A. Facchetti, Chem. Mater. 23, 733 (2011).
- C. Duan, F. Huang, and Y. Cao, J. Mater. Chem. 22, 10416 (2012).
- C. J. Brabec, M. Heeney, I. McCulloch, and J. Nelson, Chem. Soc. Rev. 40, 1185 (2011).
- S. Gunes, H. Neugebauer, and N. S. Sariciftci, Chem. Rev. 107, 1324 (2007).
- W. Li, L. Yan, H. Zhou, and W. You, Chem. Mater. 27, 6470 (2015).
- S. Bouzzine, A. Makayssi, M. Hamidi, and M. Bouachrine, J. Mol. Struct. (Theochem) 851, 254 (2008).
- Y. Dong, X. Hu, C. Duan, P. Liu, S. Liu, L. Lan, D. Chen, L. Ying, S. Su, X. Gong, F. Huang, and Y. Cao, Adv. Mater. 25, 3683 (2013).
- A. Farcas, I. Ghosh, and W. M. Nau, Chem. Phys. Lett. 535, 120 (2012).
- H. Yao, L. Ye, H. Zhang, S. Li, S. Zhang, and J. Hou, Chem. Rev. 116, 7397 (2016).
- S. Ozdemir, M. Sendur, G. Oktem, O. Dogan, and L. Toppare, J. Mater. Chem. 22, 4687 (2012).
- T. Yasuda, T. Imase, Y. Nakamura, and T. Yamamoto, Macromolecules 38, 4687 (2005).
- H. A. M. v. Mullekom, J. A. J. M. Vekemans, and E. W. Meijer, Chem. Eur. J. 4, 1235 (1998).
- C. P. Yau, Z. Fei, R. S. Ashraf, M. Shahid, S. E. Watkins, P. Pattanasattayavong, T. D. Anthopoulos, V. G. Gregoriou, C. L. Chochos, and M. Heeney, Adv. Funct. Mater. 24, 678 (2014).
- M. Moral, A. Garzon, G. Garcia, J. M. G. Roldan, and M. F. Gomez, J. Phys. Chem. C 119, 4588 (2015).
- R. Dutta and D. J. Kalita, Eur. Phys. J. D 71, 87 (2017).
- A. Kurowska, A. S. Kostyuchenko, P. Zassowski, L. Skorka, V. L. Yurpalov, A. S. Fisyuk, A. Pron, and W. Domagala, J. Phys. Chem. C 118, 25176 (2014).
- M. Wang and F. Wudl, J. Mater. Chem. 20, 5659 (2010).
- G. R. Hutchison, M. A. Ratner, and T. J. Marks, J. Am. Chem. Soc. 127, 2339 (2005).
- R.-M. Wu, H.-X. Liu, M.-Z. Ao, D.-Y. Wu, and X. Wang, Comput. Theor. Chem. 1046, 107 (2014).
- J. Cornil, D. Beljonne, J.-P. Calbert, and J.-L. Bredas, Adv. Mater. 13, 1053 (2001).
- R. A. Marcus, Angew. Chem. Int. Ed. Engl. 32, 1111 (1993).
- N. S. Hush, Electrochimca. Acta 13, 1005 (1968).
- R. A. Marcus, Rev. Mod. Phys. 65, 599 (1993).
- J. Yin, K. Chaitanya, and X.-H. Ju, Comput. Theor. Chem. 1062, 56 (2015).
- Y. H. Park, Y.-H. Kim, S. K. Kwon, I. S. Koo, and K. Yang, Bull. Korean Chem. Soc. 31, 1649 (2010).
- R. Srivastava and L. R. Joshi, Phys. Chem. Chem. Phys. 16, 17284 (2014).
- M. Malagoli and J. Bredas, Chem. Phys. Lett. 327, 13 (2000).
- Y. Li, L.-Y. Zou, A.-M. Ren, and J.-K. Feng, Comput. Theor. Chem. 981, 14 (2012).
- K. Sakanoue, M. Motoda, M. Sugimoto, and S. Sakaki, J. Phys. Chem. A 103, 5551 (1999).
- B. C. Lin, C. P. Cheng, and Z. P. M. Lao, J. Phys. Chem. A 107, 5241 (2003).
- S. Chai and J.-D. Huang, Comput. Theor. Chem. 1069, 48 (2015).
- V. Coropceanu, J. Cornil, D. A. d. S. Filho, Y. Olivier, R. Silbey, and J.-L. Bredas, Chem. Rev. 107, 926 (2007).
- C. Zhao, W. Wang, and Y. Ma, Comput. Theor. Chem. 1010, 25 (2013).
- A. Touhami, R. B. Chaabane, and A. Allouche, Comput. Theor. Chem. 1073, 123 (2015).
- C.-H. Chen, C.-H. Hsieh, M. Dubosc, Y.-J. Cheng, and C.-S. Hsu, Macromolecules 43, 697 (2010).
- M. Kuik, G.-J. A. H. Wetzelaer, H. T. Nicolai, N. I. Craciun, D. M. D. Leeuw, and P. W. M. Blom, Adv. Mater. 26, 512 (2014).
- R. Dutta and D. J. Kalita, Comput. Theor. Chem. 1132, 42 (2018).








