
Designing a highly ordered topological structure is an interesting methodology to explore new functional materials with fantastic physiochemical properties. Covalent organic framework (denoted as COF) is a class of rapidly developed crystalline porous materials constructed from the organic molecules with particular groups (such as boronic, amino and formyl groups).
These novel materials feature predictably deigned topologies, highly ordered and permanent porosity, high surface area and low matter density. In particularly, layered two-dimensional (2D) COFs possess extended covalently conjugated 2D layers and precisely integrated columnar p-arrays with uniform and perforated one-dimensional (1D) pore channels. Therefore, COFs have shown the promising and global applications in the selective separation of gases, catalysis, organic electronic devices, photoelectrochemistry, electrical energy storage, chemical sensing and ion conduction membranes.
In order to improve the initiative insolubility, poor dispersion, uncontrolled morphologies and randomly distributed gain sizes of microcrystalline COF powders, it is attractive to improve the dispersion of COFs by miniaturizing them into nanometer scale with tunable shape and size. While controllably synthesizing well-dispersed covalent organic frameworks (COFs) with uniform both morphology and size is still a challenge. Recently, Professor Dong Wang and Dr. Bing Sun from Institute of Chemistry, Chinese Academy of Sciences (ICCAS), China reported a couple of works on the templated synthesis of highly ordered COF structure, modulating the morphologies and dispersing properties of COFs and their applications.
Template synthesis is an easy and effective methodology for preparing the well-defined nanostructures with desired shape and size and creates the possibility for various practical applications. For the template synthesis method, the surface properties of templates largely determine the synthetic procedure and quality or performance of synthesized structures. The functional groups on the surface of templates can facilitate the dispersion properties of templates and improve the compatibility of the templates to building blocks.
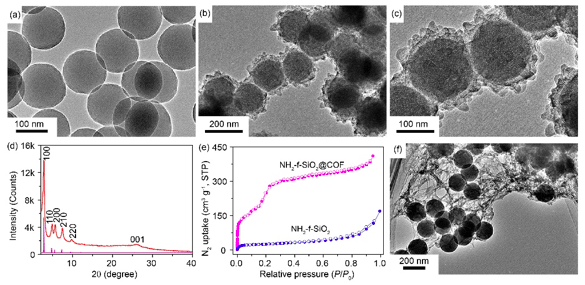
Figure 1. TEM images of (a) NH2-f-SiO2 microsphere, NH2-f-SiO2@COF in (b) large scale and (c) magnified scale. (d) PXRD pattern of NH2-f-SiO2@COF and a simulated pattern corresponding to the eclipsed conformation for COF. (e) N2 sorption isotherms of NH2-f-SiO2@COF and NH2-f-SiO2. (f) TEM image of COF growth on SiO2 without amino functionalization. Reproduced with permission from Springer Nature.
As the proof of the template synthetic concept, the template-directed synthesis of imine-based COF shell on a general template model, amino-functionalized SiO2 (NH2-f-SiO2) microspheres are first proposed. By using a convenient solvothermal method, the COF shells with high crystallinity and ordered open porous structures are synthesized (as shown in Figure 1). The thickness of the COFTTA-DHTA shell on NH2-f-SiO2 surface can be tuned by modulating the initial monomer dosages. Then, COF hollow sphere was obtained by etching the SiO2 core of NH2-f-SiO2@COF in 1% HF solution. High crystallinity and high porosity of COF shells remain. Both NH2-f-SiO2@COF and COF hollow spheres show colloidal property in the solvent.
The amino groups modified on SiO2 spheres are essential to the formation of the well-defined COF-based structures. The decorated amino groups on SiO2 microspheres can preferentially anchor the formyl monomer molecules to the surface of SiO2 templates via the Schiff-base reaction between amino groups and aldehyde groups. When another monomer molecules are slowly and continuously added, the nucleation process of COF and dynamic Schiff-base reaction between two-type building blocks preferentially occur on the template surface. Furthermore, the initial formyl molecules covalently anchored onto SiO2 surfaces serve as the docking sites to fix the growing COF around SiO2 templates. In the process of solvothermal growth and self-heal adjustment, crystalline COF shells with regular pore structures are obtained. In contrast, the weak interactions between SiO2 surface without amino functionalization and grown COF structures cannot maintain the formation of the ordered and stable COF shells.
This work represents a general way to prepare more COF-based structures with controllable morphology. It could also be applied to prepare COF-based heterostructure by growing COF shells on amino-modified templates. The controllable shape and size of COF may be propitious to explore their further applications in energy storage, drug delivery, and catalysis. This study Template synthesis of imine-based covalent organic framework core-shell structure and hollow sphere: a case of COFTTA-DHTA was published in Science China Chemistry (2017, 60, 1098–1102).
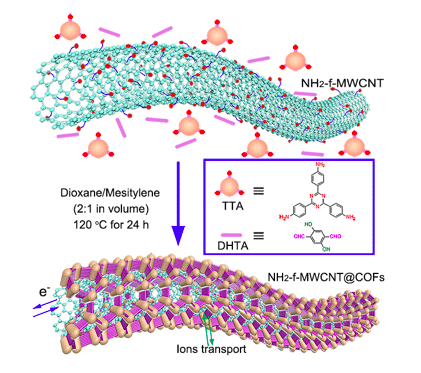
Figure 2. Schematic illustration of COFTTA-DHTA shells on the NH2-f-MWCNT surface. COFTTA-DHTA was constructed from TTA and DHTA via imine linkages. Reproduced by permission of The Royal Society of Chemistry.
The synthesis of COFs with highly ordered pore channels on amino-functionalized multi-wall carbon nanotubes (NH2-f-MWCNTs) is also proposed illustrated as in Figure 2. The synthesis of COFs on a conductive template surface, particularly carbon nanotubes (CNTs) and graphene is anticipated to provide oriented and well-defined porous COFs and also improve their electrical conductivity for further applications. The modified amino groups are also essential in the formation of highly crystalline and porous COF shells.
Coupled with the improved electrical conductivity in the presence of MWCNT skeletons, NH2-f-MWCNT@COF is developed as a capacitive electrode material and shows enhanced electrochemical capacitance. The normalized specific capacitance is 11.3 mF cm-2 at 0.4 A g-1. It is larger than that of most activated carbons and some graphene or CNT-based materials. The energy density and power density of the NH2-f-MWCNT@COF-based electrochemical capacitor are evaluated to be 11.33 W h kg-1 and 272 W kg-1, respectively. During 1000 charge-discharge cycles, a specific capacitance retention of ~96% can be obtained, which indicates the cycling stability of the NH2-f-MWCNT@COF electrode.
These findings are described in the article entitled Interfacial synthesis of ordered and stable covalent organic frameworks on amino-functionalized carbon nanotubes with enhanced electrochemical performance, published in the journal Chemical Communications. This work was led by Bing Sun from the Chinese Academy of Sciences.








