
Clouds are one of the main factors influencing the atmospheric system: they are one of the most important elements in the radiative balance on Earth and are involved in the hydrological cycle, thus affecting the climate in a general way. The effect of clouds on all these processes depends on the characteristics of the particles conforming the clouds: ice crystals and/or droplets.
Cloud particles are initiated by the process called nucleation. The nucleation is called heterogeneous nucleation when a foreign particle act as a nuclei. The nucleating particles are ice nuclei particles (INPs) or cloud condensation nuclei (CCN) if they contribute to origin ice crystals or cloud droplets, respectively. Specifically regarding the formation of ice crystals, at temperatures higher than –36°C the only way to form ice particles involves the presence of INPs.
There are different modes of heterogeneous nucleation of ice (Vali, 1985). The main mode, which is involved in the formation of ice crystals in mixed-phase clouds (clouds formed by ice particles and water droplets), is the immersion freezing mode. It is produced when an INP is immersed in a droplet of water at temperatures lower than 0°C. Different aerosols can act as INPs on the immersion mode: pollen (Pummer et al., 2012), bacteria (Haga et al., 2014), fungal spores (Spracklen y Heald, 2014), biogenic particles (O’Sullivan et al., 2014), volcanic ashes (Hoyle et al., 2011), particles from biomass burning (Petters et al., 2009), etc. However, it has been shown that mineral dust particles have a dominant role as INPs by immersion freezing (Hoose et al., 2010).
The sources of natural mineral particles in the atmosphere are the arid regions. For instance, Patagonia, in Argentina, is the most important source of dust in South America (Prospero et al., 2002). The particles generated in this region can even reach Antarctica when they are transported by the wind (Gassó et al., 2010) and contribute to ice particles formation. Therefore, considering the large contribution of these particles to the aerosol mass concentration in the atmosphere and the importance of the INPs, we study the ability of these particles as INPs by immersion freezing mode.
This study was performed using natural mineral particles collected in one site placed in Patagonia. Different suspension concentrations were prepared in order to analyze the effect of these particles on the freezing temperatures of water droplets when these particles are immersed. The volume of water droplets was varied between 2.5 to 100 mL. The evolution of the drop during the decrease in the temperature was followed with a digital camera. The changes in the reflectivity and opacity of the drops during the freezing process allowed to register the freezing temperature. As an example, Figure 1 shows these changes during the freezing of a drop.

Figure 1: Evolution of the reflectivity and opacity of a drop while its freezing. (Credit: María Laura López)
The registered freezing temperatures (Tf) were analyzed as a function of the droplet volumes and the suspension concentrations. It was observed that Tf increases with the suspension concentration and the drop volume. Both trends can be explained considering the presence of active sites on the surface of INPs. The freezing process is initiated on these active sites. Given that the number of these active sites is increased when the drop volume and the suspension concentration are increased, Tf is consequently increased. This can be seen in Figure 2, which shows the trend ofthe freezing temperature regarding these both variables.
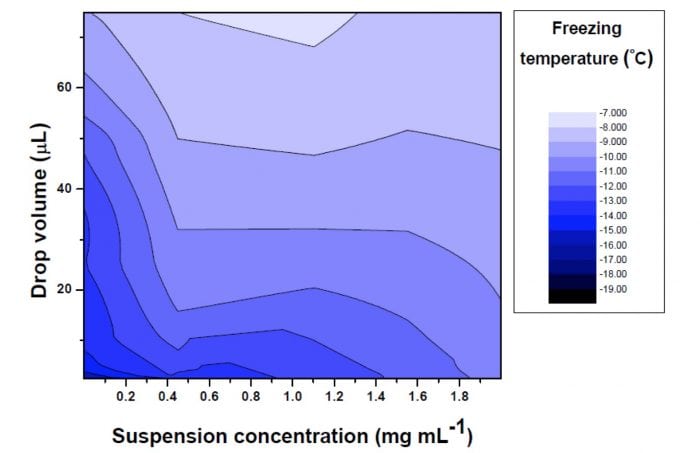
Figure 2: Three-dimensional plot representing the freezing temperature for different drop volumes and suspension concentrations. (Credit: María Laura López)
Figure 3 shows the increase in Tf when the suspension concentration is increased in drops of 10 mL. Observe the plateau in Tf at high suspension concentration. It was related to the aggregation of the particles when the suspension concentration is increased. The formation of these aggregates contributes to keep constant the number of active sites and, therefore, to keep constant Tf.
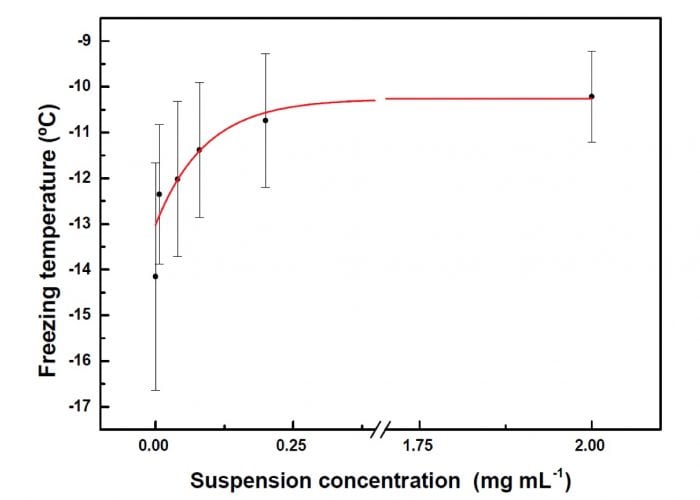
Figure 3: Freezing temperature as a function of different suspension concentrations for drops of 10 microliters. The red line corresponds to the fit from Equation 1 (see text). (Credit: María Laura López)
In order to quantify the trends in the temperature freezing, the following parameterization for Tf (ºC) versus the suspension concentration (C, in mg mL-1) was performed for each drop volume:
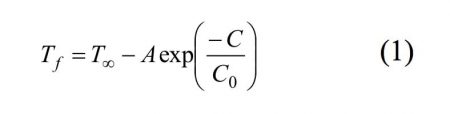
where T∞, A, and C0 are fitting parameters (this fit is also shown in figure 3). T∞ is the maximum temperature that can be reached for each drop volume by increasing the suspension concentration. The lower temperature reached for a given drop volume can be obtained from the subtraction between T∞ and A (from now on called Tmin), and the highest suspension concentration that contributes significantly to the increase in the freezing temperature can be obtained from C0. Table 1shows the corresponding fitting parameters, as well as the square of the correlation coefficient (R2). Finally, Tw and SDTw, also shown in this table, are the mean and the standard deviation, respectively, of Tf for pure water, which were experimentally obtained.
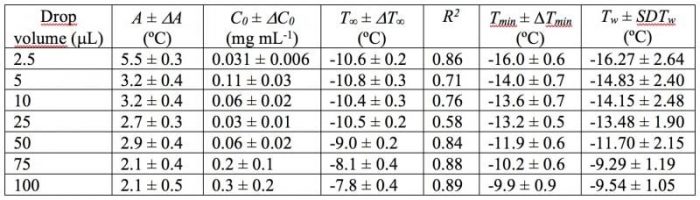
Table 1: Characteristics parameters derived from the fitting parameters for each of the drop volumes.
Observe that Tmin and Tw were very similar for every droplet volume, which indicates the good fit of Eq.1 for the experimental results. The increase in Tf when the suspension concentration is incremented can be explained because the number of particles acting as INPs is higher when the suspension concentration is enhanced. For a droplet containing more than one INP, Tf will be controlled by the INP that nucleates ice at the highest temperature. The formation of aggregates would explain the constant Tf values following certain suspension concentration and the lack of a trend in C0values.
From these results, the role of the collected particles as INPs by immersion freezing mode is shown. Considering that, due to westerly dynamics, Patagonian atmospheric dust is not effectively deposited inland but rather blown directly to oceanic waters (Gaiero et al., 2004), the result obtained in our research leads us to think that Patagonia soil, a source of dust, could affect the formation of precipitation in far away areas. This research is only the beginning. The study about the influence of mineral particles on the ability of cold clouds to form precipitation through the ice phase is the next challenge.
These findings are described in the article entitled The role of natural mineral particles collected at one site in Patagonia as immersion freezing ice nuclei, recently published in Atmospheric Research. This work was conducted by Dr. María Laura López, Laura Borgnino and Eldo Ávila from CONICET and the National University of Córdoba.
References
- Gaiero, D.M. et al. (2004): The signature of river- and wind-borne materials exported from Patagonia to the southern latitudes: a view from REEs and implications for paleoclimatic interpretations. Earth and Planetary Science Letters 219, 357-376.
- Gassó, S. et al. (2010): A combined observational and modeling approach to study modern dust transport from the Patagonia desert to East Antarctica. Atmos. Chem. Phys. 10, 8287-8303.
- Haga, D.I. et al. (2014): Ice nucleation by fungal spores from the classes Agaricomycetes, Ustilaginomycetes, and Eurotiomycetes, and the effect on the atmospheric transport of these spores, Atmos. Chem. Phys., 14, 8611-8630.
- Hoose, C. et al. (2010): A classical-theory-based parameterization of heterogeneous ice nucleation by mineral dust, soot, and biological particles in a global climate model, J. Atmos. Sci. 67, 2483-2503.
- Hoyle, C.R. et al. (2011): Ice nucleation properties of volcanic ash from Eyjafjallajökull. Atmos. Chem. Phys., 11, 9911-9926.
- O’Sullivan, D. et al. (2014): Ice nucleation by fertile soil dusts: relative importance of mineral and biogenic components, Atmos. Chem. Phys., 14, 1853-1867.
- Petters, M.D. et al. (2009): Ice nuclei emissions from biomass burning. Journal of Geophysical Research 114, D07209.
- Prospero, J.M. et al. (2002): Environmental characterization of global sources of atmospheric soil dust identified with the nimbus 7 total ozone mapping spectrometer (TOMS) absorbing aerosol product, Rev. Geophys. 40, 1002.
- Pummer, B.G. et al. (2012): Suspendable macromolecules are responsible for ice nucleation activity of birch and conifer pollen, Atmos. Chem. Phys., 12, 2541-2550, 2012.
- Spracklen, D.V. and Heald, C.L. (2014): The contribution of fungal spores and bacteria to regional and global aerosol number and ice nucleation immersion freezing rates, Atmos. Chem. Phys., 14, 9051-9059.
- Vali, G. (1985): Nucleation Terminology, J. Aerosol S., 16 (6), 575-576.









