
A woman has just recently given birth to a child born from the longest known frozen human embryo. Tina Gibson, from Eastern Tennessee, gave birth to Emma Wren Gibson on November 25th. The delivery was overseen by Dr. Jeffrey Keenan of the National Embryo Donation Center. The event may break a new record, while simultaneously raising questions about what to do with preserved embryos.
Gibson’s successful pregnancy and delivery likely beat out the previous record for a successful birth from a frozen embryo, which was an embryo frozen for 20 years. Emma Gibson weighs around 6 pounds and 8 ounces, and is 20 inches long, and seems healthy. Tina Gibson and her husband Benjamin Gibson say they don’t care whether or not it’s a world record, they are just thankful that their child is healthy. The embryo that became Emma was initially frozen back in October 1992, when Tina was only 18 months old. Tina noted that had Emma been born at the initial time, they could have easily been the right age to be friends.
However, while the Gibsons may be most excited about raising their child, from a scientific perspective the successful implantation of a 24-year-old frozen embryo is a substantial milestone and technological triumph. According to officials at the NEDC, the embryo was frozen on October 14, 1992. Yet some experts argue that it is unclear whether or not Emma’s birth is a genuine record, as companies in the United States don’t have to report the age of embryos to the government, only the outcome of the pregnancies.
The cryopreservation of embryos has been going on since at least the early 80s, and the first ever baby born from a cryopreserved embryo was born in 1984. Since then, the technology involved in preserving embryos through freezing them has advanced, and this recent success, if indeed a record breaker, shows that an embryo can be safely stored for at least two decades and still be viable. This could mean more choices and opportunities for parents who have fertility issues, and it also allows for those who suspect that they will lose their fertility (such as cancer patients) to put aside some embryos until they can successfully pursue being a parent.
There is a substantial demand for services to help women conceive, through in vitro fertilization, the use of frozen embryos, or otherwise. The Center for Disease Control and Prevention estimates that around approximately 7.3 million women, between the ages of 15 to 55, have utilized a fertility service of some kind. This accounts for around 12% of American women total.
After an egg is extracted from a donor, it is combined with sperm in a laboratory dish, which creates an embryo. The embryo is then frozen and preserved for later use. There is only is an average survival rate of 75% when unfreezing embryos. There is an approximately 25% to 30% successful implantation rate for unfrozen embryos. Fewer embryos are created now than in the past, largely because the process of in vitro fertilization is so successful. Couples who have completed their families and as such no longer need their additional embryos often donate them to facilities that put them in cryostorage.
The potentially record-breaking pregnancy is just one sign that the landscape of fertility look substantially different to what it looked like decades, or even just a few years ago. The past few years, and particularly 2017, have seen substantial advancements in fertility technology.
Earlier this year, physicians at the Children’s Hospital of Philadelphia created a synthetic womb, which enabled premature lambs to successfully survive and develop. The lambs were taken out of the womb and only 105 days after starting development (about 22 weeks) and placed in the bio-bags. While in the bio-bags they continue to develop for four weeks, developing their lungs and other organs, until they reached the point where they could survive on their own. While the technology was tested on lambs, it appears that the bio-bag successfully mimic the conditions of the human womb which means that premature babies could have a chance to continue developing in natural conditions, and it could even save the lives of severely preterm children.
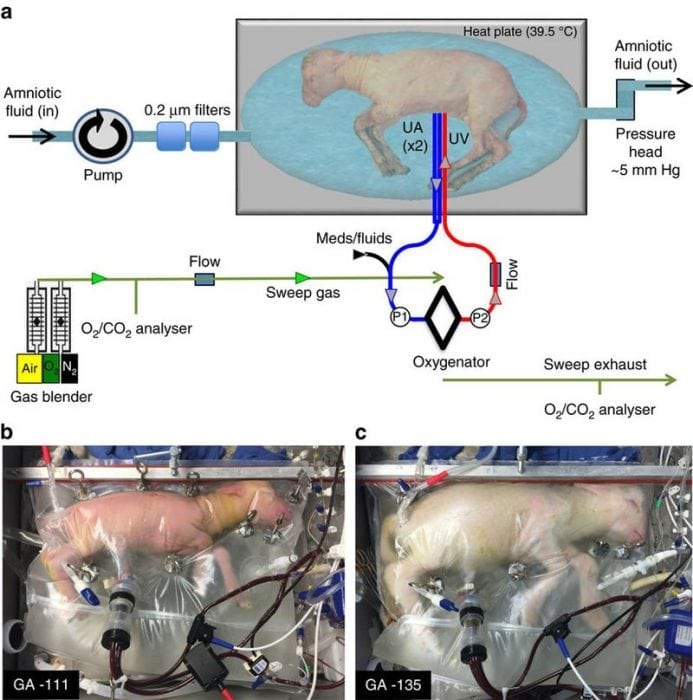
Photo: By artridge, Emily A.; Davey, Marcus G.; Hornick, Matthew A.; McGovern, Patrick E.; Mejaddam, Ali Y.; Vrecenak, Jesse D.; Mesas-Burgos, Carmen; Olive, Aliza; Caskey, Robert C.; Weiland, Theodore R.; Han, Jiancheng; Schupper, Alexander J.; Connelly, James T.; Dysart, Kevin C.; Rychik, Jack; Hedrick, Holly L.; Peranteau, William H.; Flake, Alan W. (25 April 2017). “An extra-uterine system to physiologically support the extreme premature lamb”. 8. The Author(s): 15112. Retrieved 2018-01-18. – https://www.nature.com/articles/ncomms15112?error=cookies_not_supported&code=22c9740a-44a8-4a0d-96ec-0f35ad370f1a, CC BY 4.0, https://commons.wikimedia.org/w/index.php?curid=67012621
This year also saw a woman successfully get pregnant and give birth, thanks to a transplant of an entire uterus. The transplant was carried out by the Baylor Annette C. and Harold C. Simmons Transplant Institute, and the successful transplant offers hope to the approximately 13,500 women in the US who were born without the uterus. Furthermore, approximately 15% of women may have a nonfunctioning uterus. The transplant operation also offers hope to transgender women who in the future may be able to have children.
Engineers are even working to create 3-D printed ovaries, which allow him for mice to give birth. The 3-D printed ovaries were created out of a Jell-O like substance combined with living cells to stimulate its growth and development. The gelatine-like substance was used as a base and biological principles were used as scaffolding to cement the structure into a viable ovary. The researchers hope that one day their technology could be used to enable infertile women to conceive.
Though these medical advances seem like futuristic technology, they also present themselves as real opportunities for those who want a family yet are unable to conceive naturally. New fertility technology brings along a wide variety of opportunities and benefits, but it also brings ethical challenges and questions with it. Some are worried that the increase in the viability of premature babies means that the timeline for acceptable abortions is being pushed forward, while others are concerned that the viability of frozen embryos is being utilized by religious groups to push for more restrictions on abortions. As with any technology, the debate rages on about how to best apply it in an ethical manner.









