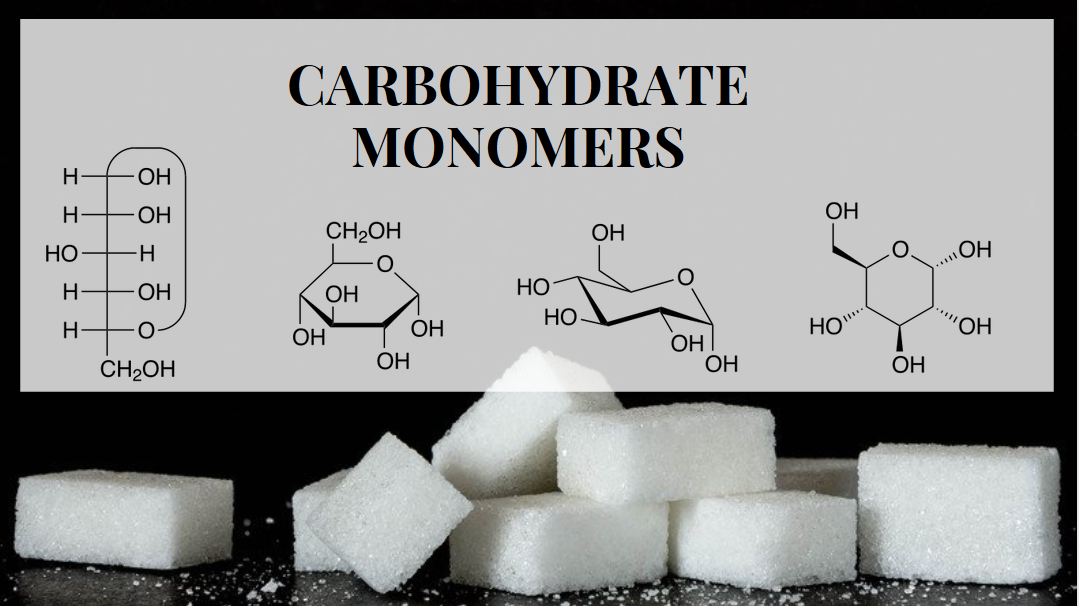
Monomers of carbohydrates are simple sugars and the basic building blocks of carbohydrates, they are also known as monosaccharides and are used by the cells of living things to store and produce energy.
What structure do monosaccharides have? How do cells use them for energy?
Defining Monosaccharides
Before delving into the finer details of monosaccharides, let’s take a moment to define them. A monosaccharide is a simple sugar, and they are also the basic units, or building blocks, of carbohydrates. Monosaccharides are forms of monomers, molecules capable of combining with other similar molecules to create more complex polymers.
“When in doubt, ingest carbs.” — Rachel Cohn
The typical chemical formula of a monosaccharide is CnH2nOn. In other words, they are created out of one hydroxyl group (OH) as well as a carbonyl group (C=O). There are various forms of monosaccharides, each defined by how many carbon atoms the molecule has. A diose molecule has two carbon atoms, a trioses molecule has three, a tetrose has four, etc. Some of the most important monosaccharides are fructose (six-carbon) and fructose (also six-carbon).
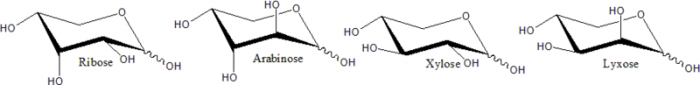
Some examples of pentose (5-carbon) molecules. Photo: Public Domain
Monosaccharides bond with each other to create carbohydrates, referred to as either polysaccharides or oligosaccharides. If an oligosaccharide has just two monosaccharides composing it, it’s called a disaccharide. The combination of more than 20 monosaccharides creates a polysaccharide, a complex carbohydrate.
The Monosaccharide Structure
Monosaccharides are created out of hydroxyl groups and carbonyl groups. The carbonyl group is made out of carbons, which are capable of forming up to four bonds. These carbon molecules bond together in various ways, and one of the carbons in the carbon chain will form a double bond with an oxygen atom. The presence of this carbon-oxygen double bond creates the carbonyl group. Note that if the carbonyl group is found at the end of the chain, it means that the monosaccharide is within the aldose family. If the carbon-oxygen double found within the middle of the chain it means the monosaccharide is part of the ketose family.
Molecules which have identical formulas yet different structures are known as structural isomers. Meanwhile, isomeric molecules that have both the same molecular formula and the same order of bonded atoms, yet have a different orientation in a 3D space are referred to as stereoisomers.
Monosaccharides that have eight or more carbon molecules are rarely observed because they’re quite unstable and break apart very quickly.
The Function of Monosaccharides
Monosaccharides play a variety of different roles within the cells of animals and plants. The function of monosaccharides is to store energy and produce energy. Most organisms derive their energy by breaking apart the monosaccharide known as glucose and utilizing the energy that is released by the chemical bonds of glucose.
Some monosaccharides are used to create fibers that come together to form various cellular structures. An example of this process is the creation of cellulose by plants. Some forms of bacteria can also create a cell wall out of a different type of polysaccharide. The cells of animals also encase themselves in a structure made out of polysaccharides, which as previously noted come from smaller monosaccharides.
Monosaccharides don’t require intestinal digestion to be absorbed, though oligosaccharides must first be hydrolyzed down to monosaccharides before they can be absorbed.
Important Monosaccharides
Three of the most important monosaccharides are three sugars known as glucose, fructose, and galactose. Each of these monosaccharides at the same chemical formula: C6H12O6. The fact that the three common sugars all have six carbon atoms means that they are all hexose molecules. While the same molecular formula is found in the three sugars, each of the sugars has a different arrangement of atoms.
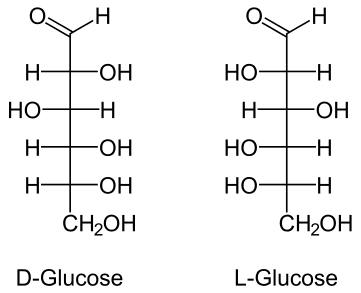
The structural layout of two glucose variations. Photo: Public Domain
Glucose
Glucose is a monosaccharide that provides both structure and energy to organisms. Molecules of glucose are broken down by the process of glycolysis, and as a result of the process, it creates both energy and chemical precursors which are used in cellular respiration. When a cell has all the energy it needs, excess glucose can be stored by the cell for later use. Glucose is stored by being combined with monosaccharides, and some plants create long chains of stored glucose – starch. This starch is then disassembled later when the plant needs energy. Animals have a similar storage method, storing glucose as polysaccharide glucose.
“The brain’s preferred source of fuel is glucose/carbohydrates. And when you go on a low-carb, high-protein diet, your brain is using low-octane fuel. You’ll be a little groggy, a little grumpy.” — Jack LaLanne
Galactose
Galactose is produced by many different organisms, but mainly mammals. Mammals have galactose in their milk, and when young mammals drink the milk they get the energy stored within it. Galactose is frequently combined with glucose to create a disaccharide known as lactose. Lactose can hold a substantial amount of energy and young mammals create special enzymes to break down the bonds of lactose. When an animal is weaned off its mother’s milk it slowly begins to cease production of the enzymes capable of breaking down glucose and galactose.
Humans are the only mammal that continues to drink milk in adulthood, and as a result it the human species has enzymes capable of continuing to break down lactose throughout their lives. Those who are lactose intolerant have problems properly digesting the lactose in milk because they lack the enzymes to do so.
Fructose
Fructose is extremely similar to glucose, though it has a different structure. While glucose has a carbonyl group at the end of the molecule, fructose has a carbonyl group in the middle portion of the chain. Much like glucose, fructose has six carbons with hydroxyl groups attached to them. Because fructose has a different ring shape to it, fructose is processed differently than glucose. Different monosaccharides need different enzymes to break them down. Much like the other monosaccharides, fructose can be combined with other monosaccharides. When fructose combines it creates oligosaccharides. An example of this is sucrose, which is made out of one fructose molecule connected to a glucose molecule.
There are exceptions to general rule that monosaccharides follow as CnH2nOn formula. One exception is the monosaccharide Deoxyribose, which has the chemical formula: H−(C=O)−(CH2)−(CHOH)3−H
2-deoxyribose has a critical role in biology, as it is part of the molecule known as deoxyribonucleic acid (DNA), the building blocks of life.









