
Potential applications of graphene in bioanalytic devices and functional composites have recently attracted wide attention. For these applications, graphene often needs to be exposed to, or in intimate contact with, aqueous systems containing ions and bio(macro)molecules.
Understanding the surface structure of graphene in aqueous media is, therefore, important. Several studies on the interactions between biomolecules and graphene and its derivatives in aqueous media have been reported. However, the methods employed in these studies provided limited structural information on the adsorbed molecular layer at the buried graphene-water interface.
Synchrotron X-ray reflectivity (XRR) is a quantitative technique for unraveling the structural information at such buried interfaces.1,2 Our recent paper3 reported unprecedented results on the structure of the same batch of graphene samples in air, using XRR along with several complementary methods, further demonstrating the feasibility of XRR for probing the graphene interfacial structure. In this study,4 synchrotron XRR (European Synchrotron Radiation Facility beamline BM28; energy 14 keV) has been applied to investigate the CVD graphene on SiO2 (widely used by the research community for graphene properties and applications), when graphene was immersed in water and phosphate buffered saline (PBS) at 25-60 °C. Contact angle measurements have also been performed to assess the wettability of the graphene, and atomic force microscopy (AFM) imaging was attempted for complementary topographic information.
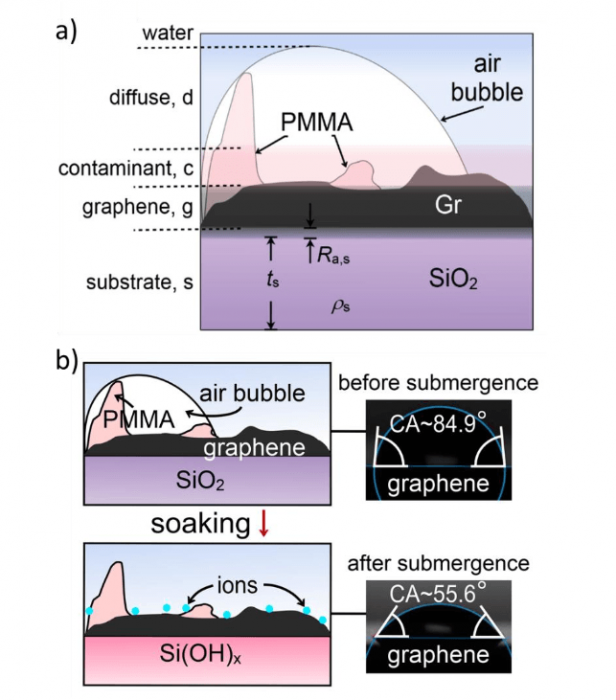
Fig. 1. a) Physical model used to fit the XRR curves of graphene in water includes a diffuse air-bubble layer atop a contaminant layer on graphene. b) The water contact angle (CA) of graphene before and after its submergence in Milli-Q water for 24 h. Figure republished with permission from Elsevier from https://doi.org/10.1016/j.carbon.2018.10.093
Fitting the scattering length density (SLD) to the XRR curves on graphene indicated the presence of a diffuse air-bubble layer (SLD ρd = 6.72×10-6 Å-2, thickness td = 84.9 Å, and roughness Ra,d = 31.9 Å) on top of graphene upon initial immersion in water (Fig. 1a). In contrast, AFM imaging provided inconclusive topographic information due to the intrinsic experimental difficulties, but the results were consistent with an inhomogeneous interface.
Interestingly, the diffuse air-bubble layer diminished after the graphene was submerged in water for 24 h at 25 °C. This is also evident from its enhanced wettability, with the water contact angle on graphene decreasing from 84.9±0.4° to 55.6±0.4° after submergence (Fig. 1b). Furthermore, an additional 10.1 Å nanolayer appeared atop graphene after soaking at room temperature. The thickness of this nanolayer increased to 11.8 Å as graphene was heated 60 °C in PBS buffer, and was retained upon cooling back to the room temperature. We attribute this nanolayer to ion adsorption on graphene from PBS (Fig. 1b), a process that was enhanced by heating. Such ion adsorption would manifest as a result of a series of environmental stimulations, such as temperature-enhanced ion mobility, possible formation of a silanol layer on the SiO2 substrate, and water structure disruption at higher temperatures at the graphene/water interface.
These unprecedented results point to the complex interfacial structure of graphene in contact with aqueous media, and possible structural evolution in response to the surroundings. Such information is relevant to bioanalytic and nanotechnological applications of graphene in which its structure at the interface between water and electrolyte solutions is an important consideration to the efficacy and functionality of the devices or the processes.
These findings are described in the article entitled Graphene surface structure in aqueous media: Evidence for an air-bubble layer and ion adsorption, recently published in the journal Carbon.Acknowledgment: The XRR measurements were performed at XMaS beamline (BM28), the UK-CRG, European Synchrotron Radiation Facility (ESRF), 6 Rue Jules Horowitz, BP 220, 38043 Grenoble CEDEX 9, France. The project was partially supported by Household Care Analytical, Procter & Gamble Newcastle Innovation Centre Whitley Road, Longbenton, Newcastle NE12 9TS, UK.
References:
- Briscoe, W. H., Speranza, F., Li, P. X., Konovalov, O., Bouchenoire, L., van Stam, J., Klein, J., Jacobs, R. M. J. & Thomas, R. K. Synchrotron XRR study of soft nanofilms at the mica-water interface. Soft Matter 8, 5055-5068 (2012)
- Speranza, F., Pilkington, G. A., Dane, T. G., Cresswell, P. T., Li, P. X., Jacobs, R. M. J., Arnold, T., Bouchenoire, L., Thomas, R. K. & Briscoe, W. H. Quiescent bilayers at the mica-water interface. Soft Matter 9, 7028-7041 (2013)
- Zhou, L., Fox, L., Włodek, M., Islas, L., Slastanova, A., Robles, E., Bikondoa, O., Harniman, R., Fox N., Cattelan, M., Briscoe, W.H. “Surface structure of few layer graphene”, Carbon 136, 255-261 (2018)
- Zhou, L., Islas, L., Taylors, N., Robles, E., Bikondoa, O., Briscoe, W.H. “Graphene surface structure in aqueous media: Evidence for an air-bubble layer and ion adsorption”, Carbon 143, 97-105 (2019)









