
Metamorphosis in the animal kingdom involves choreographing a complex series of cell rearrangements, cell deaths, and cell divisions. In insects, discrete cell populations that are set aside early in development are destined to form many adult (imago) structures such as wings, eyes, legs, and antennae.
These imaginal discs remain quiescent and undifferentiated until they are prompted by specific hormonal titers to begin adult differentiation.
Within each abdominal segment of larval Manduca, however, cells destined to form adult cuticular (exoskeleton) structures are not segregated into discrete groups of imaginal discs; but these presumptive adult cells are not only intermixed with cells that form larval and pupal cuticular structures but also contribute to the formation of those abdominal cuticles. The cuticle and underlying epithelial monolayer of cells that produce it are collectively referred to as integument.
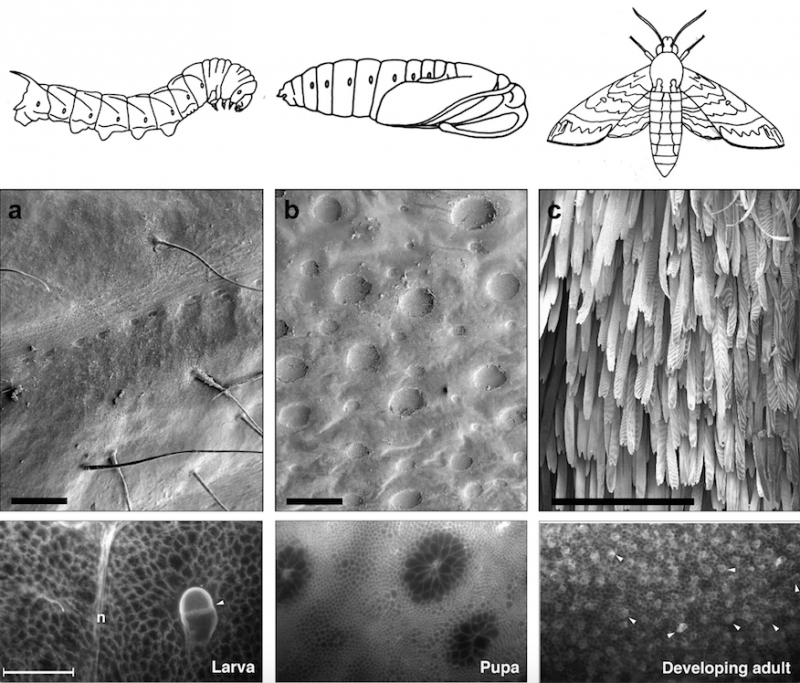
Figure 1. Larva-pupa-adult metamorphosis of the moth Manduca (top panel), its abdominal cuticles (middle panel), and the cells that produce these cuticles (bottom panel). Bar in bottom panel = 100 µm. Republished with permission from Elsevier from: https://doi.org/10.1016/j.ydbio.2018.03.017.
In the top panel, the dorsal abdominal segments (tergites) of the last larva instar (left), pupa (middle), and adult moth (right) of Manduca sexta transform over a 25-day period. In the middle panel, the cuticle of the wandering larva (day 0) has characteristic long sensory bristles (arrows). The thick brown pupal cuticle (b) has characteristic invaginated patches of cuticle (pockmarks), and the thinner cuticle of the newly-emerged adult on day 25 (c) has characteristic densely-packed scale cells.
In the bottom panel, the covert developmental events that occur at different stages in cells of the underlying epithelial monolayer reflect these differences in the larval, pupal, adult cuticles.
The epithelial cells that produce these cuticles undergo striking changes in shape and size during the larva-pupa-adult transformation. Differences in larval, pupal and adult cell shapes and sizes are revealed by antibody labeling of cell membranes. The largest pair of larval cells represents one of the conspicuous sensory bristles (trichogen) and its associated socket cell (tormogen) of the larval integument. This bristle/socket cell pair is indicated with an arrowhead. The large cells of the larval monolayer and pupal monolayer either transform or disappear by the developing adult stage when cells of uniform size and uniform shape occupy the tergite monolayer. At the developing adult stage, the arrowheads point to the cells that will form the future scales of the adult moth (scale mother cells).
The larger cells epithelial cells of the abdominal segments are known as polyploid cells. The larger cells have undergone several DNA duplications without cell divisions; these cells are known as polyploid cells. A normal diploid cell contains two sets of chromosomes (2C), whereas these polyploid cells contain from 4 to 128 times the normal diploid complement of DNA.
At larva-pupa apolysis when cells have detached from their old larval cuticle but not yet formed their new pupal cuticle, these polyploid cells are free to move and rearrange. Most cells rearrange to minimize their contacts with surrounding cells by forming invaginated circular rosettes within the confluent epithelial monolayer. At pupa-adult apolysis, when the cells are again free of their old pupal cuticle but have not yet formed their new adult cuticle, the large polyploid cells within these rosettes as well as some surrounding polyploid cells are programmed to die, to be extruded, and to be replaced by cells of uniform size and DNA content (ploidy).

Figure 2. Metamorphosis progresses between pupation (left panels) and the beginning of adult development (right panels); top panels show shapes and sizes of nuclei; in the bottom panels bright spots label nuclei of those cells that are programmed to die after pupation as adult-specific structures begin forming. Striking changes in cell size, shape and arrangement occur during metamorphosis of the abdominal monolayer during this three-day period. At pupation (left panels) cells are still attached to their pupal cuticle. At a stage two days later (middle panels) these cells retract from their pupal cuticle; large numbers of polyploid cells are destined to die. Three days after pupation, cells begin forming adult-specific structures (right panels). Bars in bottom panel = 100 µm. Republished with permission from Elsevier from: https://doi.org/10.1016/j.ydbio.2018.03.017.
In the epithelial monolayer of this pupal moth abdomen, polyploid cells segregate in circular patches from cells of equal or lower ploidy. These circular configurations of polyploid cells with high ploidy levels (pock-mark cells) in the midst of cells with same-sized or smaller nuclei are presumed to arise by sorting of epithelial cell types according to their differences in affinity. Cells of a given affinity would minimize their contacts with cells of a different affinity by forming a circular interface with those cells.
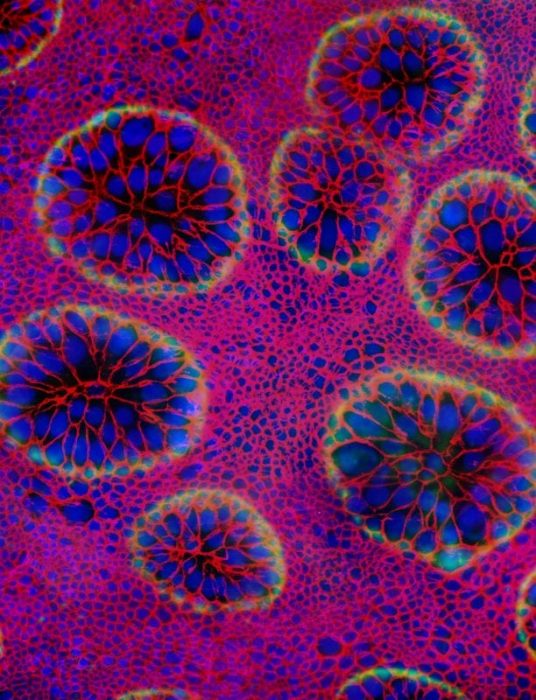
Figure 3. (color). Cells of these circular patches in the pupa are destined to disappear three days later at the beginning of adult development – i.e. when cells retract from the pupal cuticle and begin differentiating as adult cells (pupa-adult apolysis). These cells whose nuclei are labeled blue and whose membranes are labeled red secrete an overlying cuticle (green). Discrete invaginations (pockmarks) of overlying cuticle whose edges are highlighted in green overlie each of the circular aggregations of polyploid cells. Republished with permission from Elsevier from: https://doi.org/10.1016/j.ydbio.2018.03.017.
These findings are described in the articled entitled, Remodeling of the abdominal epithelial monolayer during the larva-pupa-adult transformation of Manduca, recently published in the journal Developmental Biology. This work was conducted by James B. Nardi, Charles Mark Bee, and Catherine Lee Wallace from the University of Illinois.









