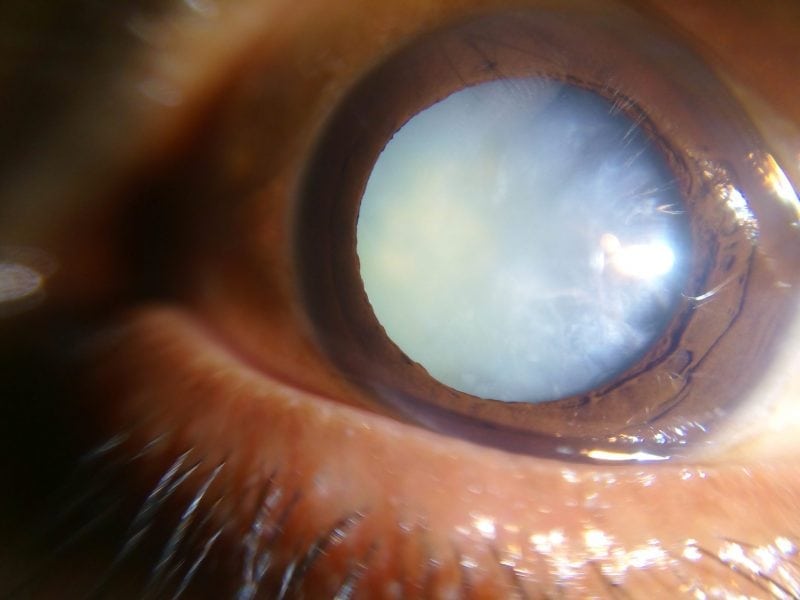
Millions of individuals undergo cataract surgery each year. Although removal of cataracts restores vision, approximately 20% of adults and most children develop a re-clouding of the lens called a secondary cataract or posterior capsule opacification (PCO). Normally, the posterior portion of the capsule surrounding the lens lacks cells that would interfere with light on its way to the retina. PCO develops when cells accumulate on the posterior capsule and contract to produce wrinkles that affect vision. A laser is used to treat PCO; however, some patients experience side effects from the procedure, and treatment is not available worldwide.
Cells with the potential to give rise to contractile muscle were identified in the lenses of multiple species, including humans. These “Myo/Nog” cells were originally discovered in the early chicken embryo by their synthesis of the skeletal muscle-specific transcription factor MyoD and noggin that inhibits potent molecules called bone morphogenetic proteins. Myo/Nog cells are incorporated into the eyes and other organs during early stages of development. The release of noggin from Myo/Nog cells is essential for normal embryogenesis. Embryos lacking Myo/Nog cells do not form skeletal muscle, the nervous system is grossly abnormal, and the lens and retina are either missing or misshapen.
Myo/Nog cells are also present in numerous organs of the adult, including the eyes and skin. While rare in normal tissues, they increase in number in response to injury and migrate to wounds. In the diseased retina of rodents, Myo/Nog cells protect neurons and reduce vision loss. A different role for Myo/Nog cells was discovered in human lens tissue removed during cataract surgery. Wounds in the lens tissue were surrounded by Myo/Nog cells that had differentiated into skeletal muscle-like, contractile cells called myofibroblasts. The emergence of myofibroblasts in cultures of human lens tissue was completely prevented when Myo/Nog cells were specifically eliminated with 3DNA nanocarriers containing doxorubicin, a cytotoxin commonly used for cancer chemotherapy.
The next step was to demonstrate that Myo/Nog cells were associated with PCO in the eye itself. To this end, their behavior was examined in ocular tissues of rabbits that develop clinically significant PCO within a month after cataract surgery. Similar to the human and mouse eye, Myo/Nog cells were found in low numbers in the lens and ciliary body of the rabbit eye. Contraction of the muscle within the ciliary body causes a change in the shape of the lens that focuses light on the retina. The force of contraction is transmitted to the lens via the fibers of the zonule of Zinn. In all three species, Myo/Nog cells were observed on the zonule fibers.
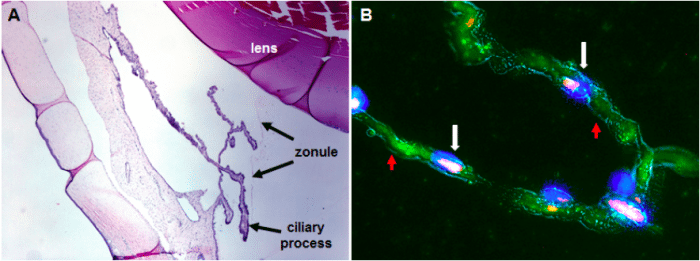
Figure 1. Myo/Nog cells are present on the zonule of Zinn. A. A tissue section of the rabbit anterior segment was stained with hematoxylin and eosin. The zonule fibers extend from the processes of the ciliary body to the lens. B. Myo/Nog cells were labeled with the G8 antibody (red). Their nuclei were stained with Hoechst dye (blue). The zonule fiber was labeled with an antibody to fibrillin (green). (Credit: Jacquelyn Gerhart)
A day after cataract surgery in rabbits, Myo/Nog cells had increased in the lens and ciliary body and their numbers remained elevated for four weeks. Myo/Nog cells were migrating on the zonule fibers between the ciliary body and lens. Coincident with the development of PCO, Myo/Nog cells had populated the posterior capsule, differentiated into myofibroblasts and overlaid prominent wrinkles.
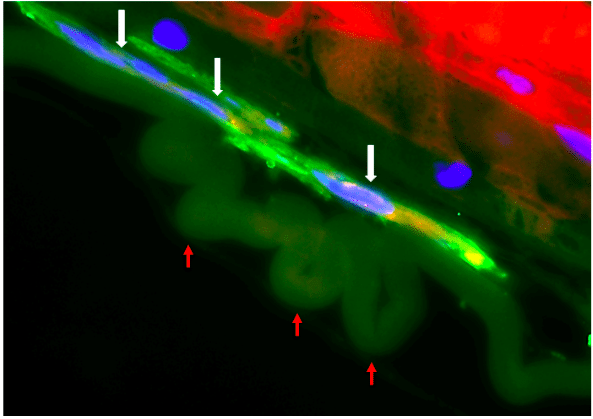
Figure 2. Differentiated Myo/Nog cells contract to produce wrinkles in the lens capsule. Myo/Nog cells labeled with Hoechst dye (blue) and antibodies to G8 (red) and the muscle protein alpha-smooth muscle actin (green) (white arrows) overlaid wrinkles in the lens capsule (red arrows). (Credit: Jacquelyn Gerhart)
Prevention of PCO is an important goal in ophthalmology that when achieved will reduce health care costs and preserve vision. Injection of a sustained delivery formulation of the drug that eliminates Myo/Nog cells during cataract surgery is envisioned to remove the source of myofibroblasts within the lens and target those cells that may repopulate the lens by migrating from the ciliary body on the zonule fibers.
These findings are described in the article entitled Myo/Nog cells are present in the ciliary processes, on the zonule of Zinn and posterior capsule of the lens following cataract surgery, recently published in the journal Experimental Eye Research. The authors of the paper are Jacquelyn Gerhart, Colleen Withers, Colby Gerhart, Arturo Bravo-Nuevo, Victoria Schienfeld, and Mindy George-Weinstein from the Philadelphia College of Osteopathic Medicine, Liliana Werner and Nick Mamalis from the University of Utah, Paul FitzGerald from the University of California, Davis, and Robert Getts from Genisphere, LLC.









