
Rhizobium rhizogenes is a bacterium that infects wounded plant stems. The infection results in the development of roots at the wound site (figure 1). These roots differ from normal plant roots by not growing towards the soil and by carrying many root hairs, hence their name of “hairy roots” (HRs). HRs can be cut off an infected plant and grown in a liquid culture medium in flasks or in Petri dishes (figure 2). Unlike other plant tissues which are grown in vitro, they have an indefinite growth without any phytohormone.
These peculiar characteristics of HRs are due to the expression of a few bacterial genes in the genome of HR cells. These bacterial genes are integrated into the genome of a plant cell at the moment of infection by the bacterium. Each infected cell then develops into a HR. When a foreign gene is added to the bacterium, it can also be transferred to the infected plant cell and expressed in the developing HR, which can then be propagated on large scale.
The expression of the foreign gene in HRs will result in the production of the foreign protein. This HR system has been used to produce a number of proteins, some of them of human origin. Unlike transgenic plants that are generally grown in soil and produce pollen or seeds which can convey the transgene into the environment, transgenic HRs are fully contained in vessels and would be unable to grow outside. They are as such, a safe alternative to transgenic plants.
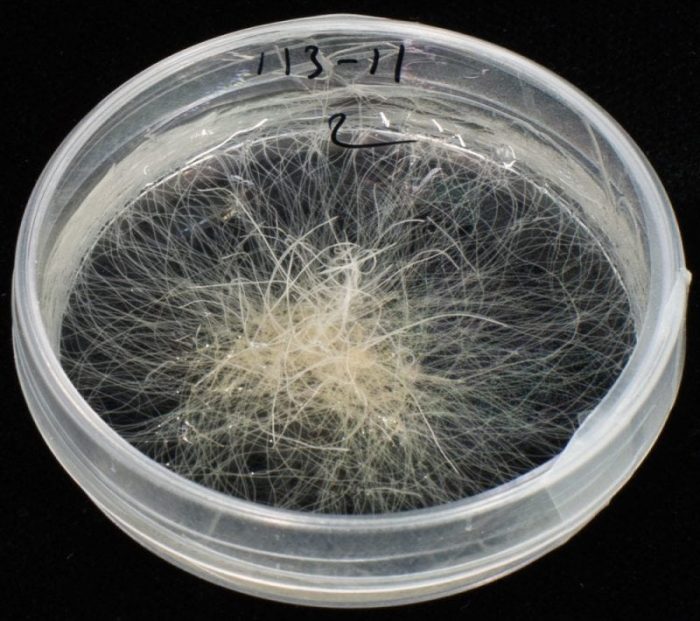
Figure 2: Hairy roots grown in a liquid medium in a Petri dish. (François Guerineau)
Lipases are enzymes that degrade lipids in the gastrointestinal tract. Some people lack pancreatic lipase because of a mutation they carry in the gene encoding the enzyme. This deficiency can result in various diseases, such as cystic fibrosis. The patients are given supplements of pancreatic lipase extracted from animals. Animal products carry the risk of transmitting infectious elements such as viruses or prions. An alternative to using enzymes from animals as therapeutic supplements in humans is to have the enzymes produced by plants. This is what this study is aimed at. The human gastric lipase was chosen because it can sustain and be active in the acidic stomach fluid and it can efficiently complement pancreatic lipase deficiency.
Previous work in our lab had shown that turnip and Arabidopsis, both members of the Brassicaceae family of plants, were valuable species for generating HRs producing foreign proteins. A refinement of the system was to get the protein secreted into the HR culture medium. This was achieved by fusing the foreign gene with a short DNA sequence encoding a signal peptide that directed the protein towards the secretory pathway of the cells. The HR system was first assessed using a jellyfish green fluorescent protein as a model.

Figure 3: Jellyfish green fluorescent protein secreted in the culture medium of hairy roots treated with 2,4-D. (François Guerineau)
The fluorescent protein was abundant in the medium of HRs expressing the gene (figure 3) and it was found that adding 2,4-D to the culture medium resulted in the presence of more protein in the culture medium. 2,4-D is an herbicide that has a strong hormone action in plants. When HRs were treated with a low dose of 2,4-D, little bumps developed on them (figure 4). These new tissues contributed to the production of the foreign protein at a high level.
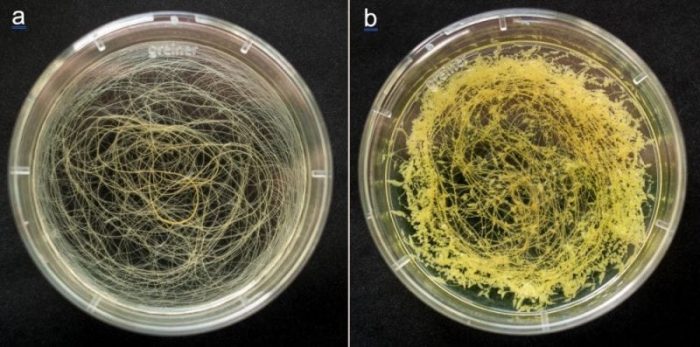
Figure 4: The development of hairy roots when untreated (a) or treated with 2,4-D (b). (François Guerineau)
HRs containing the human gastric lipase gene were then produced. Some lipase activity was measured in the culture medium of these roots. When treated with 2,4-D, they produced over twice as much lipase as did untreated HRs. As the little bumps on HRs resembled calli that often develop on plant tissues treated with hormones, we proposed naming this new HR-based protein production system “rhizocalli.”
These findings are described in the article entitled Enhanced production of recombinant human gastric lipase in turnip hairy roots, published in the journal Plant Cell, Tissue and Organ Culture (PCTOC). This work was led by François Guerineau from the Université de Picardie Jules Verne.









