
Many communities within the United States are experiencing a decline in both the quality and the quantity of water available from traditional sources (e.g., rivers, lakes, and aquifers) due to a combination of effects from climate change, increased urbanization, population growth, and prolonged droughts.
As drinking water utilities plan for their future water needs, many are considering or have incorporated direct potable reuse. Direct potable reuse is the process whereby treated wastewater is transformed into a high-quality drinking water through advanced water treatment processes. Direct potable reuse employs multiple barriers, engineered storage, and extensive monitoring to protect consumer health and ensure a high-quality product.
As with traditional drinking water sources, there is concern over the presence of man-made or anthropogenic chemicals in direct potable reuse. Recently, the presence of perfluoroalkyl and polyfluoroalkyl substances, or PFASs, has received a lot of attention. PFASs are a large group of compounds used in numerous consumer and industrial applications, e.g., food packaging, stain- and water-repellent fabrics, non-stick products, and fire-fighting foams. PFASs are widely used, extremely resistant to degradation, and soluble in water. As a result, they have been detected in rivers, lakes, aquifers, treated wastewaters, and drinking waters.
In this work, we focused on the effect of advanced treatment on the removal of both perfluoroalkyl acids (PFAAs) and PFAA precursors from treated wastewaters. PFAAs are a subset of PFASs that are commonly detected in waters and a group that includes both perfluorooctanoic acid (PFOA) and perfluorooctanesulfonic acid (PFOS). This study monitored a total of ten PFAA compounds including PFOA and PFOS using well-established liquid-chromatography tandem mass-spectrometry methods.
The broad umbrella of PFASs also includes a class of compounds labeled as PFAA precursors because they can be biologically or chemically oxidized into PFAAs. Examples of PFAA precursors include perfluorooctanesulfonamide, perfluoroalkyl phosphate mono and di esters, and polyfluorinated phospinates, and other proprietary PFASs. Often these precursors are difficult to detect analytically, so researchers have developed a method to determine their presence indirectly. In this method, PFAA levels are measured both before and after reaction with a strong oxidant (hydroxyl radical) and the difference in concentration is used to infer the concentration of precursors that must have been present.
Direct potable reuse treatment trains can generally be divided into two groups: reverse osmosis (RO)-based treatment or ozone-biological activated carbon-granular activated carbon (ozone-BAC-GAC) treatment. In both cases, contaminants are subjected to treatment processes that have the potential to either physically remove, biodegrade, or chemically transform the contaminants present, including PFASs.
As shown in Figure 1, the influents to each of the four potable reuse sites contained both PFAAs and PFAA precursors with precursors representing 30 – 67% of the total concentration. The RO-based treatment trains were both able to remove all PFASs to below detection. However, it does so at significant economic and environmental cost. Part of this cost is the disposal of the brine solution that contains all of the contaminants rejected during RO. In contrast, ozone-BAC-GAC is significantly cheaper and does not require the disposal of brine.
Significant removal of PFAA precursors was also observed in the two ozone-BAC-GAC sites, but there was a limited change in the total concentration of PFAAs. Although not shown here, treatment produced a significant shift in the type of PFAAs present from the long-chain species, like PFOA, to shorter compounds. The slight increase seen in the second ozone-BAC-GAC site occurred because of the degradation of PFAA precursors to PFAAs during treatment.
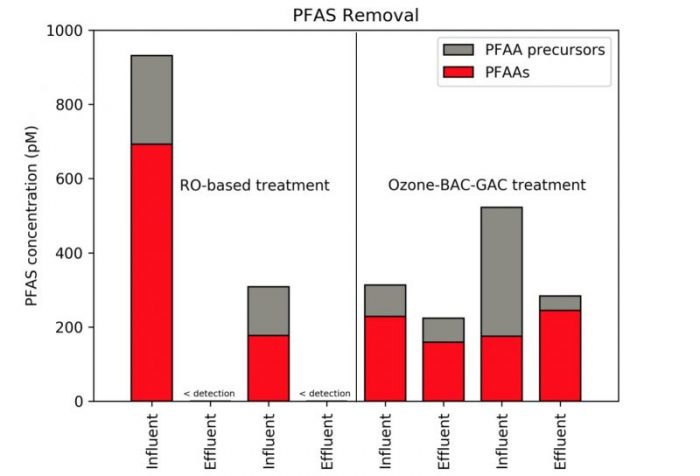
Figure 1. Removal of PFAA precursors and PFAAs in RO-based treatment and ozone-BAC-GAC treatment. The influent is the treated wastewater entering the potable reuse facility and the effluent is the finished product water. The PFAAs shown here are a molar sum of ten individual compounds. The PFAA precursors molar concentrations were generated using an oxidation method. Image modified from Elsevier from https://doi.org/10.1016/j.watres.2018.07.018.
Important takeaways from this study:
- PFAA precursors were detected in all treated wastewater samples.
- RO removed PFAAs and PFAA precursors to below their detection limits.
- Ozone-BAC-GAC favorably removed PFAA precursors and long-chain PFAAs over short-chain PFAAs.
- The sum of all PFAAs detected in final effluent product waters were not above the U.S. EPA health advisory level of 70 ng/L for the sum of PFOA and PFOS.
These findings are described in the article entitled Removal of perfluoroalkyl and polyfluoroalkyl substances in potable reuse systems, recently published in the journal Water Research. This work was conducted by Oscar Quiñones, Research Chemist, and Eric R.V. Dickenson, Project Manager, of the Water Quality Research and Development group at the Southern Nevada Water Authority.









