
Particulate Matter (PM) or aerosol is an important constituent of the Earth’s atmosphere and varies significantly in concentration, size distribution and chemical composition in ambient air.
In general, PM can be classified on the basis of their aerodynamic diameter and ability to affect the human respiratory system and body tissues into various fractions: Coarse PM (PM10), Fine PM (PM2.5) and Ultrafine PM (PM1). The coarse PM fraction, PM10, has an aerodynamic diameter of ≤10 µm and gets trapped in the upper respiratory tract while the fine and ultrafine PM fractions (aerodynamic diameter: ≤2.5 µm for PM2.5 and ≤1 µm for PM1) get retained deep into the alveolar region and cause inflammation and other cardiovascular problems.
As a result of its large-scale implications on both environment and health, atmospheric air pollution from fine PM has become a worldwide concern. Aerosols cause scattering and absorption of terrestrial and solar radiation, visibility degradation, the formation of clouds and fog and carcinogenic and non-carcinogenic toxicity, morbidity, and cardiopulmonary mortality.
Various chemical components like Organic Carbon (OC), Elemental Carbon (EC), Polyaromatic Hydrocarbons (PAHs), inorganic and organic ions (Na+, K+, Ca2+, Mg2+, NH4+, Cl–, NO3– and SO42-, HCOO–, CH3COO–) and metals (Al, As, Ba, Cd, Cr, Pb etc.) associated with PM2.5 are, in principle, responsible for all the above mentioned climatic changes and health effects. Among other chemical components of fine PM, researchers from various parts of the world report that exposure to different metals released from various anthropogenic or natural sources may lead to acute or chronic toxicity in humans.
The Indo-Gangetic Plain (IGP), a densely populated region of North India, is particularly under the influence of high aerosol loadings and has become a suitable hotspot to study atmospheric air pollution caused by fine particles (PM2.5) in terms of chemical composition, sources and health effects. During winter, mainly in the months of December, January and February, meteorological conditions like low temperature, wind speed and boundary layer height and high relative humidity become more favorable for the accumulation of pollutants near the ground surface. As a result, the levels of PM2.5 and metals released from local sources or long-range transport are enhanced and hence, affect the local air quality and are a cause of potential risk to the lives of inhabitants living in North India.
The conditions further worsen during fog occurrences in the months of December and January when the temperature is as low as 3°C to 13°C, wind speed is < 2m/s and relative humidity is >84%. During such episodes, high levels of both PM2.5 and metals released from various sources like biomass burning, vehicular and industrial emissions, soil dust and other anthropogenic activities affect the lives of both adults and children and cause toxicity in them which may be either carcinogenic or non-carcinogenic or even both.
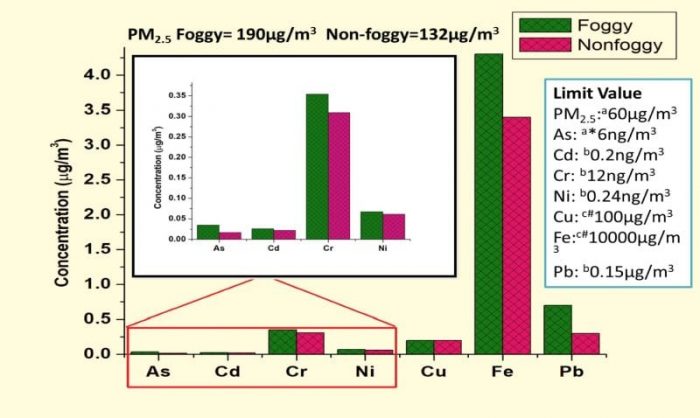
Concentrations (µg/m3) of PM2.5 and heavy metals during a foggy and non-foggy period at the suburban site of Agra and their permissible limits in ambient air by regulatory agencies (Credit: K. Maharaj Kumari)
The National Ambient Air Quality Standards (NAAQS) defines the limit value of PM2.5 to be 60µg/m3 in India. However, PM2.5 levels exceeding this value have been observed in winter at a sub-urban site of Agra with PM2.5 concentrations as high as 279 µg/m3 during foggy days and 159 µg/m3 during non-foggy days. A similar pattern of enhancement in concentrations is also observed for PM2.5 bound metals during a foggy period in comparison to the non-foggy period and the values are all above the standards laid by various regulatory bodies or agencies.
As a result of this enhancement in the concentration of metals during foggy and non-foggy periods, toxicological risk has been evaluated in the suburban atmosphere of Agra using the human health risk assessment model adapted from USEPA Integrated Risk Information Database (IRIS) and International Agency for Research on Cancer (IARC). According to the model, residential exposure of heavy metals may occur via inhalation of these metals emitted from various sources. According to the IARC and IRIS classification of metals as carcinogens and non-carcinogens, Ba, Co, Cu, Mn, Pb, and Zn were considered as non-carcinogens and As, Cd, Cr, and Ni were carcinogens.
Carcinogenic risk is evaluated in terms of Incremental Lifetime Cancer Risk (ILCR) defined as the probability of developing cancer over the course of a lifetime (assumed to be 70 years) while non-carcinogenic risk is evaluated in terms of Hazard Quotient (Hq) or Hazard Index (HI) defined as a ratio of the potential exposure to the substance and the level at which no adverse effects are expected.
The results show that both adults and children are prone to the adverse effects of metals more during foggy days in comparison to the non-foggy days and that among all the metals Cr has the maximum carcinogenic effect and exceeds the limit value of 10-6 to 10-4 set by the United States Environmental Protection Agency (USEPA) while Mn exhibits the maximum non-carcinogenic effect and exceeds the limit value of Hq=1 (see Fig.2). The toxicity value of all other metals was below the safe limit defined by the USEPA. The effect of these metals may be observed at the local level where the residents living nearby are in direct contact with the sources from where these metals may be emitted.
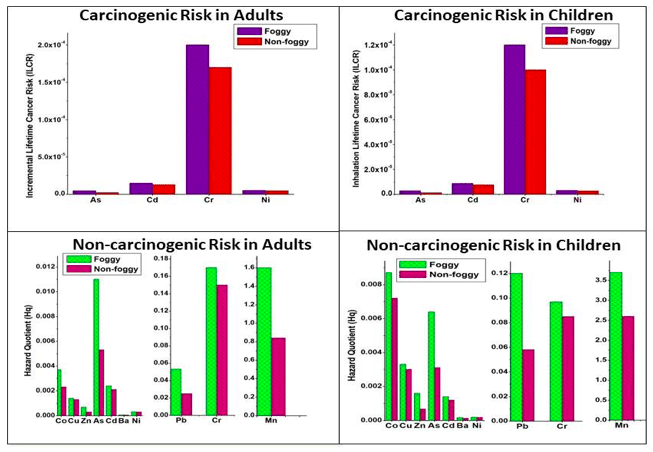
Fig. 1: Carcinogenic and Non-carcinogenic risk in adults and children during a foggy and non-foggy period. (Credit: K. Maharaj Kumari)
These findings are described in an article entitled Characterization, sources and health risk analysis of PM2.5 bound metals during foggy and non-foggy days in sub-urban atmosphere of Agra, recently published in the journal Atmospheric Research. This work was conducted by Awni Agarwal, Ankita Mangal, Aparna Satsangi, Anita Lakhani and Prof. K. Maharaj Kumari from the Dayalbagh Educational Institute.









