
Nitric oxide (NO) is a gaseous compound composed out of a single nitrogen atom and a single oxygen atom. It is the simplest of the nitric oxides (compounds containing nitrogen and oxygen) and has a molar mass of 30.1 g/mol. Nitric oxide is an important signaling molecule in vertebrate organisms and is one of the main components of acid rain. In highly concentrated amounts, it is toxic to humans and can cause serious medical issues.
Nitric oxide is a free radical, in the sense that it contains an unpaired valence electron. This unpaired valence electron makes nitric oxide very reactive and prone to forming chemical bonds. A Lewis structure of a compound is a pictorial representation of the atoms in a compound, their bonds, and the distribution of valence electrons. A Lewis structure for NO would look like:

Nitric oxide is composed of a single nitrogen atom that is bonded to a nitrogen atom. The double bar between the two chemical symbols (=) means that nitrogen and oxygen share a double bond—2 pairs of electrons. Lastly, there is a single unpaired electron on the nitrogen atom. This free radical explains the majority of nitric oxides chemical behavior.
Lewis Structures: A Quick Intro
A Lewis structure (also called Lewis dot formulas, Lewis dot structures, or electron dot structures) are pictorial diagrams that represent the bonding between atoms in a compound and the placement of electrons. Lewis structures were first introduced in 1916 by Gilbert Lewis and have been adopted as standard fare in high school and college chemistry courses ever since.
Lewis diagrams contain 3 basic elements: symbols that represent individual atoms, dots that represent electrons, and unbroken lines that represent shared electron pairs. Lewis structures are meant to show the electrons configuration and the character of covalent bonds in a compound.
Each element is represented by its chemical symbol on the periodic table. The configuration of the electrons is shown by the pattern of dots surrounding the chemical symbols. Shared pairs of electrons are written as a single line (—) and unshared electron pairs are drawn as a lone pair of dots. The total number of dots in the diagram is equal to the sum of the number of valence electrons of the atoms in the compound. So, a Lewis structure of water (H2O) would have a total of 8 electrons, one for each of the 2 hydrogen atoms and 6 for the single oxygen atom.
In general, when forming bonds, electrons will be rearranged until each atom in the compound is surrounded by 8 electrons. The tendency for atoms to acquire 8 valence electrons is known as the octet rule. Thus, when placing electrons in a Lewis diagram, one should first arrange them so that each atom is surrounded by 8 electrons. The lone exception to the octet rule is hydrogen (H). Hydrogen atoms will bond until they have only 2 valence electrons, corresponding with a complete 1s subshell. Pairs of dots that are shared between atoms are normally replaced with a single unbroken line to represent a bonded pair.
“Nothing exists except atoms and empty space; everything else is opinion.” — Democritus
If all the valence electrons are distributed and all the atoms still do not have 8 valence electrons, electrons will rearrange to form double and triple bonds between atoms. The doubling and tripling up of electron pairs allows electron-deficient atoms to gain more valence electrons while keeping the terminal atoms with 8 valence electrons.
Rules For Constructing Lewis Structures
Lewis structures are a useful tool for visualizing the electron configuration of compounds. Lewis structure for most main group 2-7 compounds can be constructed according to a set of rules:
Step 1. Identify the total number of valence electrons in the diagram
The first step is to figure out how many electrons your diagram should have. The total number of electrons in the diagram should be equal to the total number of valence electrons of the individual atoms. So, using NO as our example, the total number of electrons in our Lewis structure of NO should be equal to the sum of the valence electrons of nitrogen (N) and oxygen (O). Nitrogen has 5 valence electrons and oxygen has 6 valence electrons, so in total, our diagram should have 6=5=11 electrons.
“I, a universe of atoms, an atom in the universe.” — Richard Feynman
Step 2. Sketch a skeleton of the atomic structure.
Now we have to figure out how the atoms are arranged. If the compound is diatomic (contains 2 atoms) then the structure will just be a straight line. In compound with three or more atoms, there will be a central atom that is bonded to multiple atoms. In general, the central atom will be the least electronegative element of the compound.
In our case, NO is diatomic, so the skeletal structure will just be the 2 symbols next to each other:
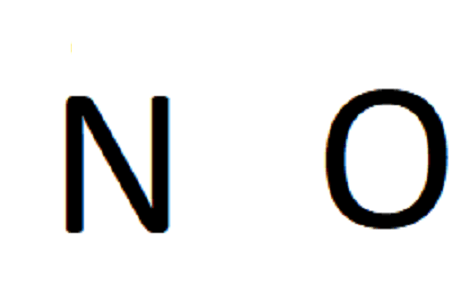
Step 3. Place a single bond between each pair
Next up is to start placing the electrons. Go through the diagram and place a single line between each bonded atom in the compound. Atoms make covalent compounds by sharing electrons, so each atom must share at least one pair with another atom.
In our case, we just need a single line between N and O:
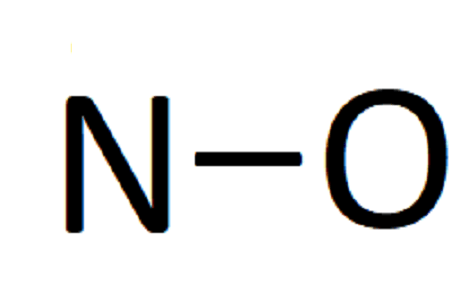
As each line represents 2 electrons, counting the total number of lines, multiplying by 2 and subtracting from the total number of valence electrons give the number of remaining electrons. Thus, we have 11−2 = 9 electrons left to place in our diagram.
Step 4. Staring with the more electronegative element, place electrons until each atom is surrounded by 8 electrons.
Atoms will try to fill their electron shells until they have a complete valence shell of 8 electrons. In a compound, the more electronegative element will pull harder on electrons, so those shells tend to be filled first. Remember, hydrogen (H) is special and only gets 2 valence electrons.
In our case, oxygen is more electronegative than nitrogen, so we fill the open slots on oxygen first:
Filing the oxygen took 6 electrons, so now we have 3 left to place. Placing the remaining electrons gives us:

Step 4. If all atoms still do not have a full shell of 8 electrons, move electron pairs to form double and triple bonds.
Sometimes there are not enough electrons for every atom to have a single bond and 6 other electrons surrounding it. In these cases, an electron will move to form double and triple bonds so that less electronegative elements can satisfy the octet rule as close as possible.
In our case, all the electrons have been placed but nitrogen only has 5 electrons. so, we move a pair of electrons from oxygen to form a double bond between the nitrogen and oxygen:
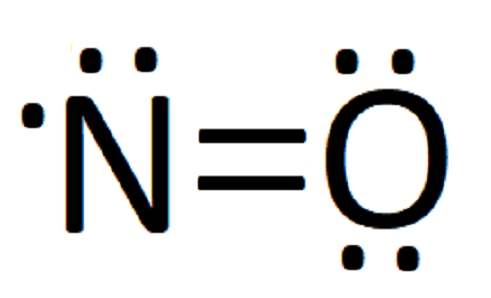 The presence of a double line (=) means that the two atoms share two pairs of electrons. In this case, we can only move one pair of electrons to give nitrogen a total of 7. If we tried to move another, nitrogen would have a total of 9 valence electrons, which would violate the octet rule. Thus, we have gotten as close as possible to giving both atoms a full octet of electrons, and our diagram is complete.
The presence of a double line (=) means that the two atoms share two pairs of electrons. In this case, we can only move one pair of electrons to give nitrogen a total of 7. If we tried to move another, nitrogen would have a total of 9 valence electrons, which would violate the octet rule. Thus, we have gotten as close as possible to giving both atoms a full octet of electrons, and our diagram is complete.
So, our completed Lewis structure for NO looks like:

Nitric Oxide As A Compound
Nitric oxide is a free radical—i.e. it has a lone unpaired electron. This lone electron gives nitric oxide an unstable chemical profile and makes it rather reactive.
Since it is very reactive, nitric oxide will react under various conditions to form more stable nitrous oxide products. In the presence of oxygen, NO will react to form the more stable nitrogen dioxide according to the following reaction:
2NO + O2 → NO2
When heated, nitric oxide will decompose into molecular oxygen and nitrogen according to the reaction
2NO + heat → N2 + O2
Nitric oxide is also one of the main causes of acid rain. Nitric oxide that is released in the atmosphere will react with hydroperoxy (HO2) to make nitrogen dioxide. Nitrogen dioxide will then react with a hydroxyl radical (OH) to form nitric acid (HNO3). Nitric acid is a corrosive compound that can damage metals and vegetation, so it can be a danger for industry and agriculture.
Limits Of Lewis Structure
“Life is not found in atoms or molecules or genes as such, but in organization; not in sybmiosis but in synthesis.” — Edwin Grant Conklin
Lewis structures are a useful way to visualize the distribution of electrons in a compound. However, they are not without their shortcomings. Specifically, more complex compounds made out of group 3-12 transition metals often do not follow the octet rule and will instead bond so that each atom has 12 valence electrons. This aberrant behavior of the transition metals is due to the delocalized electron configuration of metal compounds. Likewise, sometimes a single compound can be described by more than one Lewis structure. In these cases, the overall Lewis structure is considered a combination of the different Lewis structure, sometimes called a resonance structure.
In addition, Lewis structures do not tell you about the 3-dimensional geometry of a molecule. A molecule’s 3-d geometry explains a lot of its physical and chemical properties, like its polarity and intermolecular bonding behavior. VESPR theory is a branch of chemistry that answers questions relating to the 3-d shape of compounds and how those shapes are determined by the atoms’ bonding behavior.
Additionally, Lewis structure does not explain how atoms share electrons. This is a question reserved for molecular orbital theory. MO theory describes the behavior of atomic and molecular orbitals and how they combine in virtue of the wave-like properties of electrons.









