
During the last two decades, air pollution has been regarded as one of the toughest challenges facing human beings. As the key indicator of air pollution, PM2.5 received great public concern. Exposure to PM2.5 could cause many adverse effects, such as allergies, diseases, or even death. Furthermore, it was reported that a 10 μg/m3 increment in PM2.5 was associated with a 1.04% increase in the risk of death.
Because of the variation of composition and concentration of toxic substances adsorbed in the particles, the toxicity of PM2.5 showed obviously regional and seasonal differences. Until now, oxidative stress and inflammation were regarded as the most accepted mechanism to PM2.5-induced toxicity. Both transition metals and organic compounds contributed to ROS production and pro-inflammatory cytokines release. In cells, there are two pathways to supply energy, named glycolysis and oxidative phosphorylation. When the external environment changed, cells may rewrite the energy metabolic phenotype to adapt to the new circumstances and suppress damage.
In this study, we found that exposure to PM2.5 could change energy metabolic phenotype from more effective oxidative phosphorylation to less-effective glycolysis. Mass spectrometry-based metabolomics was used to investigate the overall metabolic changes and relevant toxicological pathways. Consequently, purine metabolism, arginine and proline metabolism, glutathione metabolism, TCA cycle, and glycolysis were affected. The biological indices determination also showed that oxidative stress and inflammation played important roles in energy metabolism disorder.
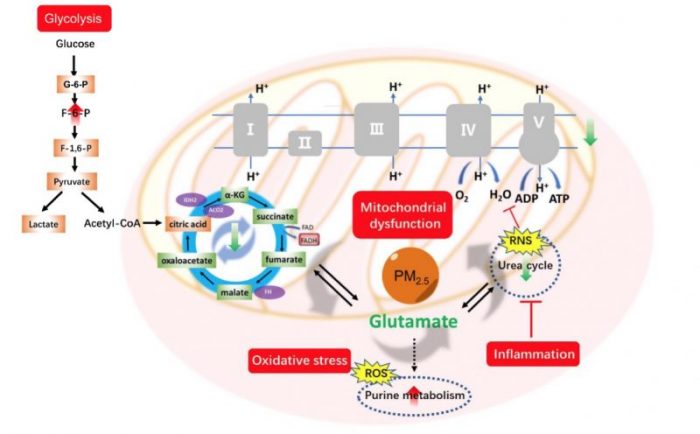
Schematic overview of the disturbed metabolic pathways in BEAS-2B cells upon PM2.5 exposure. Molecules marked in red represented the up-regulated metabolites and in green represented the down-regulated metabolites. Credit: Zongwei Cai
The metabolites in the TCA cycle (e.g. succinate, malate) were found down-regulated, which indicated that the precursors for oxidative phosphorylation were reduced. The metabolites in glycolysis (e.g. lactate, fructose 1,6-bisphosphate) were up-regulated, which proved the promotion of glycolysis in cells. Interestingly, we found the change of TCA cycle and glycolysis were also influenced by other pathways.
Purine metabolism and glutathione metabolism have been reported to be closely related with oxidative stress. The level increase of metabolites within purine metabolism indicated the production of ROS, while excessive ROS could cause mitochondrial damage, which affected the supplement of energy. Arginine and proline metabolism have been found to be controlled by inflammation. On one hand, the release of TNF-α could increase the expression of iNOS to generate nitric oxide in cells, and the accumulation of nitric oxide could compete with oxygen to reduce mitochondrial respiration. On the other hand, the conversion of arginine to urea cycle was inhibited, while some metabolites in urea cycle are the precursors of TCA cycle, so the decrease of urea cycle aggravated the reduction of TCA cycle.
In summary, the PM2.5 exposure caused the metabolic reprogramming and mitochondrial dysfunction by perturbing purine metabolism, arginine and proline metabolism, glutathione metabolism, TCA cycle, and glycolysis caused by oxidative stress and inflammation. Because endogenous metabolites are the terminal products of gene expression, metabolomics could be regarded as a powerful tool to provide the most “functional” information at the final level.
These findings are described in the article entitled Mass spectrometry-based metabolomics reveals the mechanism of ambient fine particulate matter and its components on energy metabolic reprogramming in BEAS-2B cells, recently published in the journal Science of the Total Environment. This work was conducted by Yuanyuan Song, Yanhao Zhang, Juntong Wei, Wei Chen, and Chi Kong Arthur Chung from the Hong Kong Baptist University, Ruijin Li from Shanxi University, and Zongwei Cai from the Hong Kong Baptist University and the Guangdong University of Technology.









