
Human activities have been increasing CO2 concentration (among other greenhouse gases) in the atmosphere for about 250 years, and we are already suffering from its effects on a global basis. The most direct of these effects is a worldwide increase in average global air temperature; global circulation models predict a rise of between 1.5 °C to more than 5 °C by the year 2100. Oceans are affected too; global warming is expected to raise sea surface temperature by 1 °C to 7 °C in the same period of time.
The rise in temperature in the oceans affects marine life and ecosystems, but it is accompanied by another problem caused by an increase of CO2: we call it ocean acidification. Atmospheric CO2 dissolves and reacts with seawater, forming the chemical compounds that we call the carbonate system. Essentially, CO2 and water react to form carbonic acid, which quickly dissociates into bicarbonate, releasing a proton. This bicarbonate ion can dissociate into a carbonate ion with the release of another proton. These reactions are reversible and in equilibrium, being the main source of the capability of seawater to keep its pH stable. As we humans have managed to increase the amount of CO2 in the atmosphere, more of this gas dissolves in seawater, changing the chemical equilibria of the carbonate system.
The most noticeable of these changes is a decrease in seawater pH: mean pH used to be about 8.2, but this value now is below 8.1. A further decrease of approximately 0.3-0.5 units is predicted before the end of the 21st century, displacing the seawater pH towards the acidic end of the pH scale (the pH scale goes from 0 to 14, being values < 7, acidic, and > 7, basic): hence, ocean acidification. Also, the relative concentrations of the different compounds forming the carbonate system change with pH, with an overall reduction of carbonate ions and an increase of bicarbonate ions and CO2.
Ocean acidification potentially affects all marine life, from phytoplankton to fish, but especially calcifying species (coral, echinoderms, mollusks, calcareous algae…). At lower pH levels, these organisms have to spend more energy to build their structures, which can even suffer dissolution. A lot of effort has been invested in investigating the effects of climate change and ocean acidification on marine environments.
Seaweeds are key ecosystem builders in shallow-water areas. In these environments, they uptake inorganic carbon from the carbonate system pool when performing photosynthesis (raising pH) and release CO2 with respiration (lowering pH), so seawater pH is also regulated by biological activity. These environments, when rich in producers such as seaweed and seagrass, can help mitigate the effects of ocean acidification, raising seawater pH and acting as refuge areas for negatively-affected species.
We expect that the oceans of the future will have a higher CO2 concentration (lower pH) and show higher temperature so, how will these combined stressors affect macroalgae? Understanding this is key to assess how Climate Change will change coastal ecosystems, so we conducted an experiment combining the effects of two of its main stressors (pH and temperature) on some keystone macroalgal species to contribute to addressing this question.
The study was carried out in the Canary Islands, a subtropical region that has a mix of temperate and tropical characteristics. We subjected individuals from six different algal species (Cystoseira abies-marina, Lobophora variegata, Pterocladiella capillacea, Canistrocarpus cervicornis, Padina pavonica, and Corallina caespitosa) to a set of combined temperature and pH conditions, both actual and expected for the end of the century (19, 21, 23, 25 °C, pH: 8.1, 7.7 and 7.4) and estimated production and respiration of these seaweed measuring oxygen variation in seawater.

Fig. 1: Studied seaweed species. From left to right: Cystoseira abies-marina, Lobophora variegata, Pterocladiella capillacea, Canistrocarpus cervicornis, Padina pavonica and Corallina caespitosa. Credit: José Carlos Hernández
We found that the studied species were more productive in lower pH levels than under actual conditions, so ocean acidification alone enhanced their production. The explanation is that when more CO2 is added to seawater, the amount of inorganic carbon available for photosynthetic organisms is increased, so their production may be enhanced. Although the macroalgae that we studied were benefited by this situation, this effect depends on the different mechanisms used to uptake the inorganic carbon, so not all species would take advantage from this extra source of carbon.
The temperature had different effects depending on species. Some of our seaweed produced more oxygen (a proxy for primary production) when we increased temperature, while the contrary occurred with others. Lobophora variegata, an important ecosystem engineer in the studied region, was among the species benefitting from higher temperatures levels, so this alga may thrive in a future scenario with more CO2 and higher temperatures. On the other hand, Cystoseira abies-marina, another important keystone species whose population is already in recession in the study area, showed temperate characteristics and produced less at the higher temperature levels.
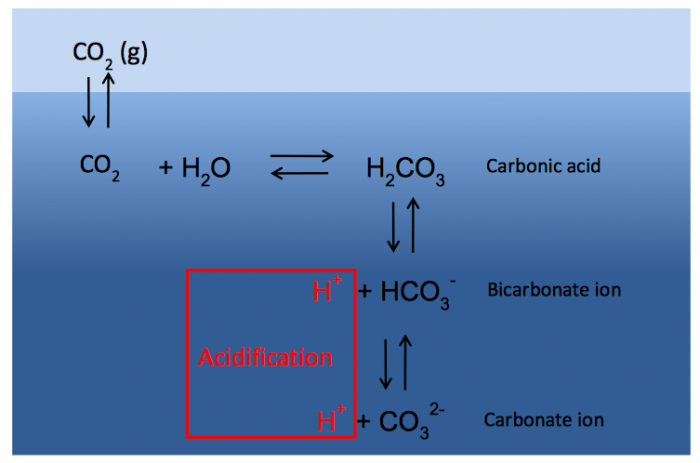
Fig. 2: The seawater carbonate system. Credit: Celso Hernández
The results that we obtained suggest that in warm-temperate regions, a future with more productive coastal ecosystems is expected, but the species composition may change towards tropical characteristics, mainly due to the increase in temperature. Temperate species may be replaced by algae that perform better at higher temperatures.
Of course, laboratory experiments like this have their limitations: restricted duration, difficulty studying the interactions between species, overlooking acclimation effects, etc. For these reasons, to get a better understanding of the effects of global warming and ocean acidification on marine ecosystems, laboratory research must be complemented with in situ experiments. In recent years, experimentation in natural CO2 vents of volcanic origin where the CO2 content in seawater is naturally higher has become an important source of information about the future oceans.
The number of CO2 vents suitable for the study of ocean acidification is limited (there are about fifteen in the world1), but one of those volcanic CO2 vents has recently been discovered in La Palma, one of the Canary Islands. This vent has the right features to be useful in ocean acidification research: it is located in shallow water areas, has the correct range of pH values, from actual to those predicted for the near future, and no toxic substances accompany the CO2 emissions2. We expect this location to become a natural laboratory that contributes to the understanding of the changes that human activities are bringing into marine environments.
These findings are described in the article entitled Macroalgal response to a warmer ocean with higher CO2 concentration, recently published in the journal Marine Environmental Research. This work was conducted by Celso A. Hernández, Carlos Sangil, and José Carlos Hernández from the Universidad de La Laguna, and Alessandra Fanai from the Università Degli Studi Di Sassari.
References
- González-Delgado S. and Hernández JC (in press). The importance of natural acidified systems in the study of ocean acidification: what have we learned? Advances in Marine Biology, 80.
- Hernández CA, Sangil C, Hernández JC (2016). A new CO2 vent for the study of ocean acidification in the Atlantic. Marine Pollution Bulletin 109, 419–426.









