Increasingly, anticancer drugs targeting mutant or fusion proteins found in a patient’s tumor are becoming the gold standard for modern-day cancer treatment. This personalized approach to oncology has led to dramatic results in the clinic, with potent tumor reduction being observed in a variety of disease traditionally considered recalcitrant to treatment. While these drugs often prove initially effective, acquired resistance invariably occurs and presents as a major limitation and problem in the treatment of cancer.
Specifically, many kinase inhibitors have been developed that target mutant or fusion forms of kinases that drive cancer through the constitutive activation of the mitogen-activated protein kinase (MAPK) signaling network. (Figure 1A) Resistance to these drugs is mediated by the reactivation of the key gatekeeper kinase MEK. While direct small molecule inhibition of MEK in combination with a variety of kinase inhibitors delays this onset of resistance, this inhibition-based strategy is only transiently effective and resistant cancers continue to prevail. Therefore, novel strategies for permanent perturbation of MAPK signaling are greatly needed.
In a paper published in Cell Chemical Biology, Peh, et al. demonstrate a general strategy to overcome MEK reactivation and cancer resistance through the utilization of MEK cleavage via procasaspe-3 activation. By combining PAC-1, a procaspase-3 activator, with targeted kinase inhibitors, the authors show a robust anticancer effect, as well as a delay and/or abolishment of resistance. This combination strategy centers around the ability of caspase-3 to cleave MEK, ultimately leading to a sustained inhibition of MAPK signaling. (Figure 1B) This exciting efficacy is observed in multiple tumor histologies, including melanoma, leukemia, and lung cancer and is also effective against cancers with driver mutations in EGFR, BRAF, EML4-ALK, and BCR-ABL proteins.
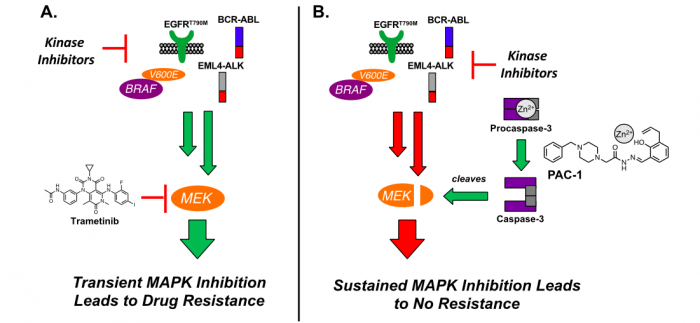
Figure 1. A. Small Molecule Inhibition of MEK in Combination with Kinase Inhibitors Leads to Transient MAPK Inhibition and Drug Resistance. B. PAC-1 in Combination with Kinase Inhibitors Leads to MEK Cleavage, Sustained MAPK Inhibition, and No Drug Resistance. (Credit: Matthew Boudreau)
Of particular interest is the ability to translate these discoveries directly into new human clinical trials. PAC-1 was discovered in Prof. Paul Hergenrother’s laboratory at the University of Illinois at Urbana-Champaign and preclinical development of PAC-1 has been the focus of a major collaboration between Prof. Paul Hergenrother and Prof. Timothy Fan, a veterinary oncologist at UIUC. The anticancer effect of PAC-1 has been explored in pet dogs with spontaneously arising tumors (canine cancer patients). This major effort has shown PAC-1 as an exciting new drug in the fight against cancer, with key success stories centered around its activity in the treatment of canine glioma.
With this efficacy in canine patients, PAC-1 is currently being explored in two human Phase 1 clinical trials: NCT02355535 and NCT03332355. Further Peh, et al. demonstrate the ability of PAC-1 to synergize with FDA approved agents: vemurafenib, trametinib, osimertinib, imatinib, and ceritinib. Therefore, the studies described by Peh and coworkers serve as a key starting point for the high impact design of future clinical trials with the ultimate goal of combating drug-resistant cancers.
These findings are described in the article entitled Overcoming Resistance to Targeted Anticancer Therapies through Small-Molecule-Mediated MEK Degradation, recently published in the journal Cell Chemical Biology. This work was conducted by Jessie Peh, Matthew W. Boudreau, Hannah M. Smith, and Paul J. Hergenrother from the University of Illinois at Urbana-Champaign.









