
Bone histology is, comparatively, a modern branch in paleontology. The techniques slowly started to be applied during the 1960s and 1970s on fossilized bones (Figure 1). But It was during the 80s and 90s that paleohistology became bigger and helped to shed light on some questions about vertebrate’s biology and evolution. Since then, over the past forty years, it is considered a reliable proxy in science providing answers that gross macro morphology cannot.

Figure 1. The image shows the final steps of the thin-section preparation. After sampling the bone and embedding it in resin, the thin-sections were wet-ground using a metallographic polishing machine with abrasive papers of increasing grit size. When a final thickness of 30–60 microns is reached, it is ready to be observed under microscope. Image courtesy Rafael Andrade
During the vertebrate’s development, as the bone grows, it changes features in several ways. Primarily, it comes from a cartilaginous precursor, once it becomes a harder tissue and bone is formed, many processes happen as the animal grows. Depositions of bone layers potentially followed by effective stops known as LAGs (Lines of Arrested Growth), matrix resorption and cavities refilling, as well as fiber orientation and vascular arrangement, can also vary. All these features, along with many others, are particularly variable among the fossil and modern taxa.
Within Crocodylia, little information is known from the bone microstructure regarding ecology, physiology, and ontogeny in fossil and modern animals. In the past thirty years, some authors have published data about the Australian freshwater crocodile, the Nile crocodile, and the American alligator. Differently, for the first time, our team could gather samples from South American caimans’ bones and piece results together. We sampled four long bones of a wild Caiman yacare; the carcass was legally collected from the Pantanal and donated for this study. Also, one sample from an unidentified fossil Caiman from the Solimões Formation was used. The aim of the study was to compare both individuals and raise comments on their growth biology.
The hind and forelimbs of C. yacare showed differences in growth dynamics. Both the femur and tibia showed four growth rings composed of a complex web of vessels and fibers. On the other hand, the humerus and radius showed simple and few vascular canals, fibers arranged in an organized manner, and three and five growth rings, respectively (Figure 2). None of the samples showed LAGs (effective stops) between the bone layers but there were, visibly, periods ranging from fast- to slow-growing tissue deposition. While analyzing the humerus of the fossil Caiman, alternately fast- and slow-growing tissues were also present, along with complex vascular canals and LAG deposition. Based on the results, the wild C. yacare seems to be a juvenile, while the fossil could be identified as an older animal due to extensive resorption cavities and active remodeling process.
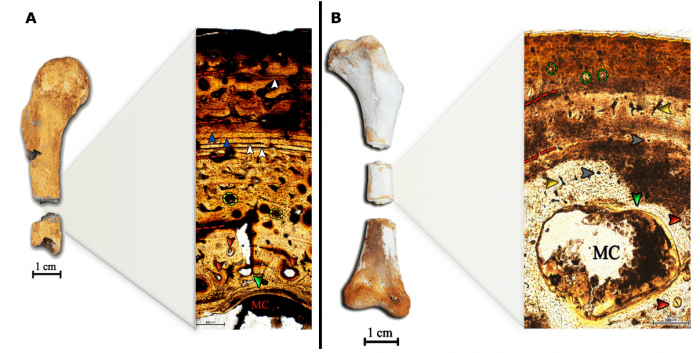
Figure 2. Image A and B show the humerus of the fossil Caiman and C. yacare, respectively. Some similarities can be observed as the cyclical growth that appears as rings and are highlighted by the dotted red lines. The growth rings show a woven bone matrix prevalence (disorganized fibers), indicating that the growth varies from fast to low deposition rates. Both specimens show the presence of medullary cavity (MC) and inner circumferential layers, indicating that the first degree of ontogenetic development was achieved by the time of death. The active remodeling process evidenced by the secondary osteons is present in both fossil and living caimans. Effective stops in bone deposition was only observed in the fossil, supporting the idea that living caimans sustain their growth during the initial stages of development. Green arrows = Inner circumferential layers; red arrows = secondary osteons; yellow arrows and dotted circles = primary vascular network; gray arrows = primary osteons; white arrows = LAGs; blue arrows = slow-growing tissue. Image courtesy Rafael Andrade
Crocodyliformes are generally known to have the capability to naturally adjust growth rates as a response to either environmental changes in the wild or even in a captive environment. Continuous and cyclical growth activity has been previously reported for Alligatoridae. Alligator mississippiensis wasn’t able to keep growing fast, even where all the environmental variables were under control and considered ideals. As C. yacare is a living species and closely related to alligators, ecological data was obtained in previous publications and then linked to the histological results that we have found in order to connect the dots. First, in the Pantanal wetlands, the rainy season ranges from October until March. This interval is considered the optimal conditions for C. yacare to grow. Oppositely, from April to September, during the drier season, the growth activity decreases. All the seasonal ecological data linked the pulses of growth observed in thin sections. In this way, we could trace the periods of high and low-deposition rates in samples bones.
Overall, comparing each feature between living and fossil specimens was limited due to the number and corresponding elements and standard samples. We concluded that Caimaninae is capable to adjust growth rates when necessary and it might be happening, at least, since the Miocene. Also, growth rates can vary within a single animal, meaning that the skeleton grows differently.









