The fossil fuel reserves of our world are depleting. Most of the government agencies are funding the research and development of fossil fuel alternatives such as bio-alcohols, biodiesel, etc, preferably produced using green technology. Ethanol, an alcohol, is produced by fermentation of sugars employing microorganisms like yeast or bacteria.The sources of sugars include food crops (sugarcane, corn, wheat, etc.) and lignocellulosic crop residues (sugarcane bagasse, corncobs, wheat straw, rice straw, etc.).
Ethanol production from food crops is very simple and does not involve energy or cost-intensive processes. However, diverting human food into fuel production is ethically wrong. On the contrary, agricultural residues are available in plenty and are even burnt to the extent of severe air pollution in most states of North India. The only hindrance to large scale lignocellulosic ethanol production is its humongous production costs, contributed by the process complexity.
Lignocelluloses are composed of lignin, cellulose, hemicellulose, inorganic, and extractive materials. Cellulose and hemicellulose constitute the sugars, which can be hydrolyzed and fermented to ethanol. But lignin encloses the hydrolysable sugars, making them unavailable for its subsequent conversion to ethanol. Therefore, conversion of lignocelluloses to ethanol involves four steps: (a) Pretreatment, where the bond between lignin and hydrolysable sugars is loosened; (b) Saccharification/hydrolysis, where simpler sugars like glucose, xylose, etc. are released from cellulose and hemicellulose; (c) Fermentation, where the sugars released from hydrolysis are fermented to ethanol by microorganisms; and (d) Purification, where the fermented ethanol is purified to improve its quality.
According to the IRENA report, the fermentation step alone constitutes 23% of the equipment costs and 16% of the total costs. Hydrolysis of pretreated lignocelluloses releases six-carbon and five-carbon sugars (hexoses like glucose, and pentoses like xylose). Baker’s yeast, i.e. Saccharomyces cerevisiae, is unable to ferment pentoses naturally. Therefore, there is the need for another yeast, like Scheffersomyces stipitis, or a bacteria, like Zymomonas mobilis, to ferment pentoses and thereby improve the yield of ethanol.
Employing two or more microorganisms for the efficient conversion of sugars is complicated because of their varying metabolic patterns, culture conditions, behavior, and interactions. For example, S. cerevisiae produces ethanol in the absence of oxygen (anaerobic condition), whereas S. stipitis requires minute concentrations of oxygen (microaerophilic condition). In the presence of oxygen, S. cerevisiae diverts its metabolism for increasing its cell biomass. S. stipitis has the ability to ferment both hexoses and pentoses; hexoses are followed by pentoses, and therefore compete with S. cerevisiae for consuming hexoses. Due to these reasons, it is difficult to predict what happens when S. cerevisiae and S. stipitis are co-cultured.
The paper “Mathematical modeling, simulation and validation for co-fermentation of glucose and xylose by Saccharomyces cerevisiae and Scheffersomyces stipitis” published in Biomass and Bioenergy in March 2018, describes the development of mathematical models to predict the concentrations of sugars, ethanol, S. cerevisiae, and S. stipitis during the co-culture of these yeasts. These models have been developed from Monod model — the simplest mathematical model to predict the growth rate of microorganisms.
In this study, mathematical models were developed considering S. stipitis’ growth rate’s dependence on dissolved oxygen concentration, in addition to other assumptions as described in the paper. Simulations and experiments were carried out simultaneously to calculate known and unknown kinetic parameters. The models were statistically validated by conducting t-test and evaluating indices of agreement. Willmott’s index of agreement for the models predicting the concentration profiles of S. cerevisiae, S. stipitis, glucose, xylose, ethanol, and oxygen was 0.867, 0.929, 0.980, 0.995, 0.910, and 0.850, respectively, satisfying the criteria for “very high” agreement between model-predicted and experimental values.
Fermentation experiments were carried out in synthetic media containing glucose and xylose. These models can be modified and used for predicting culture dynamics during lignocellulosic ethanol production. A graphical abstract of the study is given below.
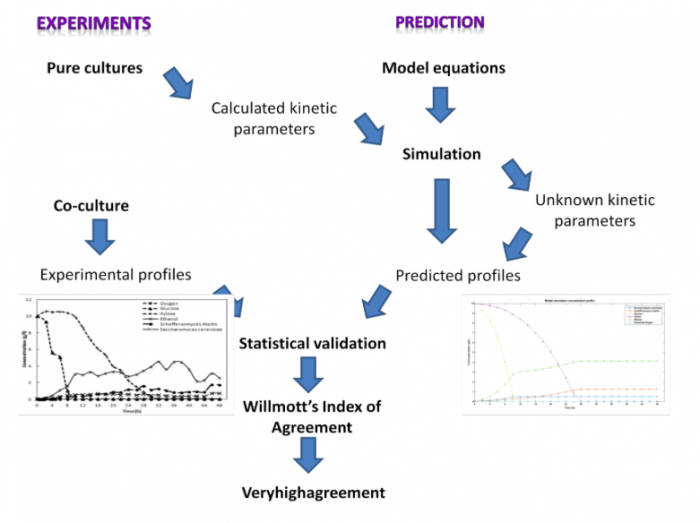
Republished with permission from Elsevier from https://doi.org/10.1016/j.biombioe.2018.01.008
These findings are described in the article entitled Mathematical modeling, simulation and validation for co-fermentation of glucose and xylose by Saccharomyces cerevisiae and Scheffersomyces stipitis, recently published in the journal Biomass and Bioenergy. This work was conducted by Siddhi Sreemahadevan, Varsha Singh, Pradip Kumar Roychoudhury, and Shaikh Ziauddin Ahammad from the Indian Institute of Technology Delhi.









