
In 2016, the world shipped over USD 220 billion of meats and seafood. Naturally, ensuring the quality and safety of these foods is a major concern for everyone in the supply chain, from the food suppliers to consumers to government agencies. In particular, monitoring the vast amounts of food can easily become a logistical nightmare. Currently, time-temperature indicators (TTIs) have been in the market for monitoring food transportation.
TTIs can indicate if the food was exposed to elevated temperatures during transportation which could signify the presence of bacterial fermentation. TTIs are a great first step, but they are far from ideal. The method monitors a time duration outside of a specified temperature rather than the actual presence of food spoilage. This is especially relevant because there have been reports where food fermentation was detected even when stored at 0 °C.
An increasingly popular method to determine food freshness/safety is testing for the presence of biogenic amine species because (i) they are produced by bacteria as they break down protein/amino acids and (ii) they can react with nitrites to generate carcinogens. Exploiting the chemical nature of biogenic amines allows us to detect their presence without large, expensive machines or complex methodology.
Because biogenic amines are basic, they can interact with dye molecules resulting in visible color change. This visualization allows for easy detection of biogenic amines/microbial fermentation of food. Furthermore, fluorescent (light-emitting) materials can further enhance detection sensitivity. Unfortunately, many conventional fluorescent dyes experience reduced light emission in the solid-state due to “aggregation caused quenching” (ACQ) (Figure 1). The chemical structures of conventional dyes are planar, which facilitate strong emission when the molecules are dissolved, but this also results in strong molecular packing in the solid-state, adversely affecting emission. In contrast, a new branch of fluorescent materials shows “aggregation induced emission” (AIE) (Figure 1). AIE molecules show fantastic emission when they aggregate and are ideal for diverse applications.
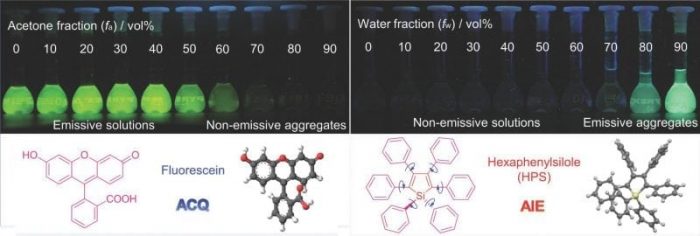
Figure 1. Conventional dye molecules are highly emissive when dissolved, but experience aggregation caused quenching (ACQ) when the molecules come together. In contrast, molecules that exhibit aggregation induced emission (AIE) are strongly emissive when they group together.
In the case of food safety monitoring, a new set of AIE molecules were designed and synthesized to allow for the easy visualization of biogenic amines (Figure 2). They combine both color change and emission turn-on (under UV light) in the presence of biogenic amines. The original dye color, orange, changes to yellow when exposed to biogenic amines and strong yellow-green emission can be observed. Because its AIE nature, the molecules can work even when deposited onto a testing strip greatly facilitating its ease of use. In this way, the strips can be sealed within food packages and alert anybody of food fermentation, which can be difficult to directly observe. Making use of the emission turn-on, the system has an incredibly sensitive detection limit of 690 ppb for ammonia, an amine species that is also generated by microbial fermentation.
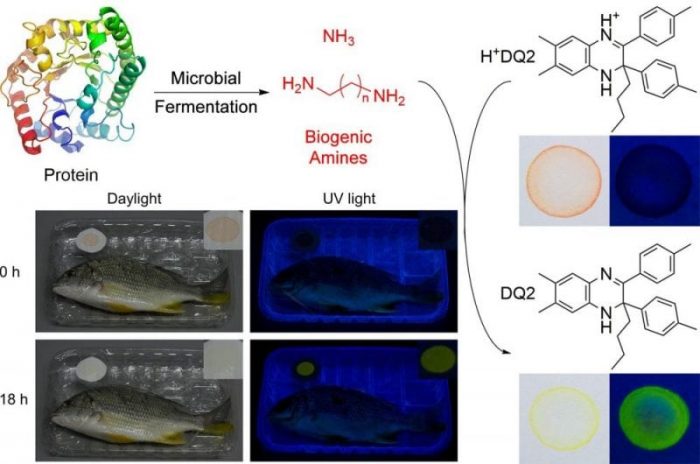
Figure 2. When proteins are broken down during fermentation by bacteria, they generate ammonia and biogenic amines. The dye, which is originally orange in color and non-emissive, changes upon exposure to the amines becomes emissive and yellow in color. The change is easily visualized. Republished with permission from Chem Eur J, a Wiley published journal.
To demonstrate their performance, the monitoring strips were used in various conditions. In the first test, monitoring strips were placed with a slice of raw salmon with one sample stored at room temperature and the other sampled stored in the refrigerator (2 °C). These two samples were also compared with a monitoring strip kept at room temperature without any salmon as a control. As refrigeration greatly hinders microbial fermentation, no changes are observed in the chilled sample.
Only the salmon sample kept at room temperature showed color change and emission turn-on. The strip only shows changes when food degradation occurs and so the control strip showed no changes. The strips were also sealed in packages of shrimp and yellowfin seabream. Within 18 hours at room temperature, although the food samples looked the same, the testing strips showed visible change indicating that fermentation had already taken place.
This study, A Highly Sensitive Bimodal Detection of Amine Vapours Based on Aggregation Induced Emission of 1,2-Dihydroquinoxaline Derivatives was recently published by Parvej Alam, Nelson Leung, Mengchen Yue, and Ben Zhong Tang in the journal Chemistry – A European Journal.









