Mapping of tissue parameters like myelin and axonal water fraction can potentially lead to evaluating myelin loss in the brain’s white matter in neurodegenerative diseases, such as multiple sclerosis and schizophrenia, and provide additional information about axonal damage, e.g. after traumatic brain injury.
Magnetic resonance imaging (MRI) is a key technology to study human brain tissue non-invasively. Ultra-high-field (UHF) MRI, performed at field strengths of 7 Tesla and above, provides increased signal and enhances image contrast and sensitivity to tissue microstructure. Well-established biophysical models allow scientists to estimate tissue parameters at the microscale in the hope of detecting pathological changes early.
Our challenge was to use model assumptions that can be tested to provide valid information about the heterogeneous brain tissue. We approached this by modeling the MRI signal from a special multiple echo gradient echo imaging experiment using several tissue compartments to relate millimeter-scale measurements (corresponding to the imaging resolution used in our experiments) to changes in tissue microstructure.
White matter can be modeled using a three-compartment model consisting of myelin, axonal, and extracellular compartments with specific, quantitative MRI tissue parameters (water fraction, relaxation time, and frequency shifts). Myelin water fraction was calculated using the ratio of myelin water fraction to the total water fraction as (myelin water fraction/ myelin +axonal + extracellular water fraction). Axonal and extracellular water fractions were also estimated in a similar manner. The myelin compartment was differentiated from the axonal and extracellular compartment based on the relaxation time. The shortest relaxation time compartment was assigned to myelin due to its higher nature of susceptibility.
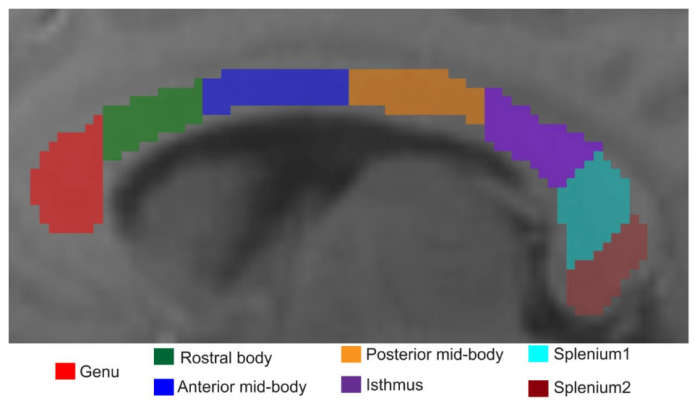
Illustration of the location of the seven regions of interest across corpus callosum which were used to study tissue characteristics via signal compartmentalization. Figure courtesy Markus Barth and Kiran Thapaliya.
As a brain region to test our white matter tissue model, we used the corpus callosum, a large white matter fiber bundle that connects the two hemispheres. We observed that across the corpus callosum, the myelin water fraction had a very similar dependence as an independently acquired histological evaluation. This is a first promising indication that we can infer relevant tissue parameters of the brain using UHF MRI and advanced modeling, potentially opening the possibility to help in the diagnosis of neurodegenerative diseases. We also observed the broader distribution of frequency shift for the myelin compartment in comparison to the other two compartments. However, the relaxation time of myelin was similar across the sub-regions of the corpus callosum. We are currently further testing our approach on clinical data sets and investigating the influence of MRI acquisition parameters, fiber direction, and model complexity to enable mapping of the whole brain.









