Adenoid Cystic Carcinoma (ACC) is a rare form of cancer originating in secretory glands, often located in the head and neck region and commonly presented in salivary glands. It is unpredictable in nature and can become very aggressive over time with metastatic spread to the lungs and other organs, such as the liver and bone. Standard of care treatments are usually tumor resection and/or radiation therapy, but they don’t always prevent the cancer from further growth. Due to treatment failure, almost 89% of the ACC patients survive the first 5 years, but the overall 15-year survival rate, unfortunately, drops to ~40%1.
For years, patient-derived xenograft-ing (PDX) in rodents has been used in cancer research to better understand the biology of a certain cancer type and to test new cancer therapies. This technique involves the implantation of a small piece of the patient’s tumor under the skin of a laboratory mouse, as sort of an incubator to let the tumor grow larger. This technique can be repeated multiple times, termed serial passaging, by using the PDX tumor to generate more tumor material in a 3D in vivo setting for experimental purposes. However, researchers have found limitations of this PDX model for certain types of cancers, including ACC. Over multiple passages, PDX ACCs showed increased tumor growth rate and changes in tumor morphology2, thus altering the original histopathology of the patient’s tumor. As the latter can skew future clinical outcomes, it was crucial for the ACC research field to find an alternative in vivo model that recapitulated the patient’s tumor.
Patient-derived orthotopic xenograft-ing (PDOX) is similar to PDX but differs in that the patient’s tumor cells are placed into a specific organ, instead of under the skin where the patient’s tumor often didn’t originate. For our new PDOX ACC model, we used the most common primary head and neck tumor location, the salivary glands (SG), to transplant the tumor cells in. Then, we questioned whether the tumors’ own glandular environment could better recapitulate the patient’s ACC and possibly lead to metastasis, as seen in the clinic. To demonstrate this, we had to confirm that our PDOX model maintained tumor fidelity, i.e. that all the characteristics of the tumor tissue we initially implanted were maintained.
We compared a) the original patient sample with b) PDX tumor tissue generated from the patient’s biopsy, and c) PDOX tumor tissue generated over multiple passages. Multiple tests were performed, including analysis of histopathological morphology, tumor growth rate, gene translocations and mutations specific to the patient’s ACC, neuronal invasion, blood vessel accumulation, and cell proliferation. We also performed a drug sensitivity test on our PDOX tumor model to validate whether this new animal model was proficient enough for testing new and validating new therapies.
What we found was very promising and exciting. ACC tumors in humans often show 3 pathological forms: solid, cribriform, and/or tubular, and are graded based on the percentage of each within the tumor. While an early transplanted PDX tumor showed a similar grade compared to our patient’s sample, PDX tumors of later passages altered their morphology and grew a lot faster; similar to the phenomena that had been seen before with other ACCs2. In contrast, our newly-generated PDOX tumor tissue grew at a constant rate over serial passaging and retained the original pathological grade of the patient’s tumor.
More importantly, we found that our ACC PDOX tumors harbored all of the original mutations of the patient’s tumor and the most commonly seen MYB-NFIB gene translocation. Preservation of these alterations is essential, as their loss may no longer mimic the original tumor, an event that often occurs in various PDX or in vitro research models.
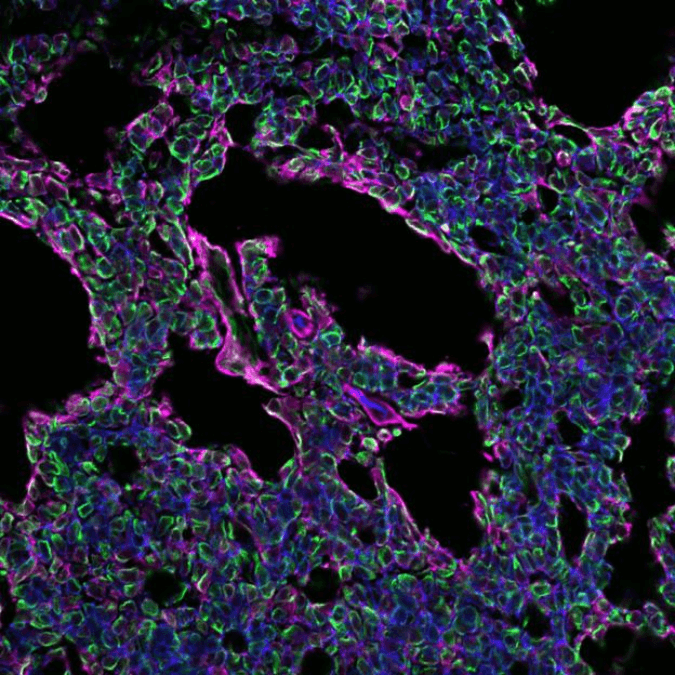
We also evaluated the level of innervation and angiogenesis within the tumor. This is important because the environment often alters to benefit tumor growth and spreading. Particularly neuronal invasion is a hallmark for ACCs. As predicted, an abundance of nerves was found in the patient’s tumor and PDOX tumors. In contrast, very few nerves were found in the PDX sample, suggesting the PDX model did not fully support perineural invasion as seen in patients.
Additionally, we found that neurotrophic factors that maintain and stimulate nerve growth were high in the PDOX and patient’s tumor but low in the PDX tumor. Angiogenesis, or new blood vessel formation, was also strikingly different between PDX and PDOX samples. The presence of blood vessels in the original human tumor were reduced in the PDX model but maintained in the PDOX model. Lastly, by delivering a drug therapy to the mouse with the PDOX tumor, we were able to slow down tumor progression, nicely illustrating that our new model can be used for future drug testing.
Overall, our research study demonstrates that our new PDOX animal model better recapitulates the patient’s tumor than the previously used PDX set-up. What does this mean for ACC patients? It opens many new doors to perform more accurate research to investigate the biology of different ACC tumor types, which has been limited due to poor models. Most importantly, it allows us to more accurately proceed with personalized medicine efforts as we can study and target a patient’s individual tumor that better reflects the clinical situation. Our PDOX model is certainly a long-awaited breakthrough for the field that fulfills the most important disparity in the head and neck cancer field.
These findings are described in the article entitled Serial patient-derived orthotopic xenografting of adenoid cystic carcinomas recapitulates stable expression of phenotypic alterations and innervation, recently published in the journal EBioMedicine.
References:
- Dillon, P.M., Chakraborty, S., Moskaluk, C.A., Joshi, P.J. & Thomas, C.Y. Adenoid cystic carcinoma: A review of recent advances, molecular targets, and clinical trials. Head Neck 38, 620-627 (2016).
- Pearson, A.T. et al. Patient-derived xenograft (PDX) tumors increase growth rate with time. Oncotarget 7, 7993-8005 (2016).









