
The International Energy Outlook 2016 (IEO 2016) published by the U.S. Energy Information Administration shows that the world’s energy consumption will grow by 48% between 2012 and 2040. And, with the increase of global warming caused by the high consumption of fossil fuels, the use of renewable and environmentally-friendly sources of energy has become crucial. Therefore, in order to alleviate the pressure on our energy supply, renewable energy storage devices (ESDs) have been extensively studied.
Lithium-ion batteries (LIBs) have achieved great success in the past decade as one of the most advanced ESDs. However, the limited resources of lithium in the earth’s crust significantly increase the fabrication cost of LIBs, which will become more and more serious as time elapses. To address this concern, ESDs based on earth-abundant and low redox-potential elements are being explored to replace LIBs. Compared to the low content of lithium in the earth’s crust (0.0017 wt %), that of potassium (2.09 wt %) is much higher. Thus, nonaqueous potassium-ion batteries (PIBs) are expected to become one of the next-generation technologies to replace state-of-the-art lithium-ion batteries in a range of stationary applications.
High-performance electrode materials are urgently desired for advanced PIBs. Current PIBs with graphite anodes, which only have an initial capacity of 230 mAh g-1 and rapidly decrease in subsequent cycles due to the large volume change during repeated insertion/extraction of K+. Metal chalcogenides could effectively mitigate this concern, due to moderate redox potential, high theoretical capacity, and good electrochemical activities.
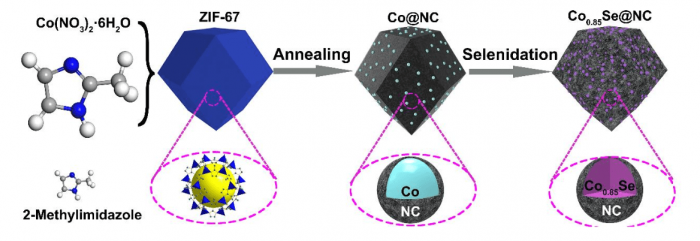
Republished with permission from Elsevier from https://doi.org/10.1016/j.mtener.2018.09.013
Herein, ZIF-67, a typical MOF, has been used as an example to prepare Co0.85Se nanoparticles encapsulated in N-doped carbon polyhedrons. N-doping of carbon not only benefits the electron conductivity as reported in the literature, but it also prevents the growth and aggregation of Co0.85Se during the preparation. This interaction is also important to electrochemical performances because it can inhibit the deformation and pulverization of active materials upon cycling. Besides N-doping in carbon, mesoporous structures indicate a large specific surface area and a good tolerance to volume change. These results enable this composite to deliver a specific capacity of 114.7 mA h g-1 at 1000 mA g-1 after 250 cycles.
The excellent electrochemical performance and environmentally-friendly synthesis approach proved that these metal chalcogenides prepared by MOFs are promising candidates for next-generation anode material for PIBs.









